3D Cell Culture
Find suitable products, scientific resources or answers to your questions within our technical library FAQ.
Checkout using your account
This form is protected by reCAPTCHA - the Google Privacy Policy and Terms of Service apply.
Checkout as a new customer
Creating an account has many benefits:
Cellular interactions in the tumor microenvironment (TME) significantly influence cancer progression and drug response. The efficacy of immunotherapies has triggered increased interest in the tumor immune microenvironment. Therefore, robust experimental systems are needed to enable researchers to model patient-specific interactions between tumor cells and immune cells.
The interaction between cancer stem cells (CSCs) and the tumor microenvironment functions as a dynamic system that fosters the generation and maintenance of CSCs, which drive tumor formation and progression. Organoids and tumoroids that recapitulate the tumor microenvironment and contain CSCs are essential tools for developing physiologically relevant tumor models for the discovery of new biomarkers, therapeutic targets, and therapeutic modalities.
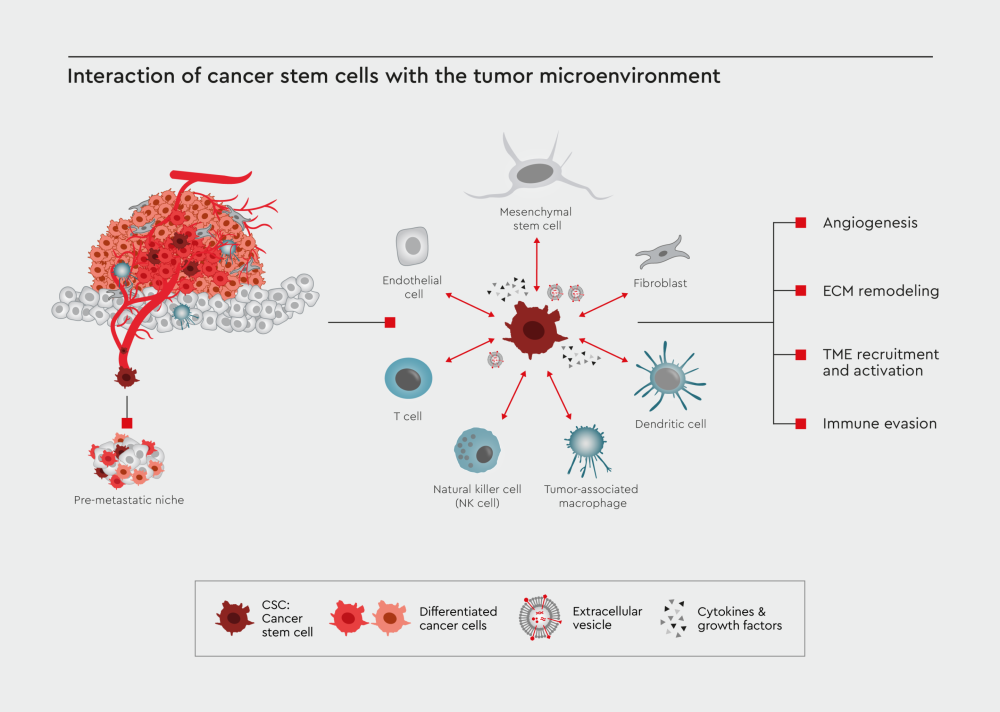

Figure 1: Interaction of CSC with the tumor microenvironment. CSCs are the main driver of tumor formation and metastasis. One of the critical components influencing the cancer progression and resistance to drug treatments is the interaction between CSCs and the tumor microenvironment, which include T cells, natural killer cells, dendritic cells, tumor-associated macrophages (TAMs), mesenchymal stem cells, fibroblasts, and endothelial cells. These cells communicate with the CSCs via secreted cytokines and growth factors or extracellular vesicles (EVs), driving the tumor progression and immune evasion.
Given the importance of CSCs in shaping the cancer microenvironment, they are promising therapeutic targets. We have developed a toolbox of cancer media that maintain a healthy CSC population within 3D tumor models.
Our media enable you to:
Our cancer media toolbox comprises the following:
Whether you want to preserve the tumor microenvironment in a tissue biopsy or construct de novo 3D models with a custom and controlled TME, it is your choice. Our cancer media toolbox empowers you to maintain a healthy CSC population and build 3D tumor models.
Let us introduce some examples of how to employ these media in the following sections.
Developing assays with physiologically relevant TME supported by CSCs
There are many ways to set up organoid cultures embedded in the tumor microenvironment. Here, we describe an example of a workflow for constructing a de novo organoid model as a standardized and reproducible model for drug testing using our cancer cell media (Figure 2).
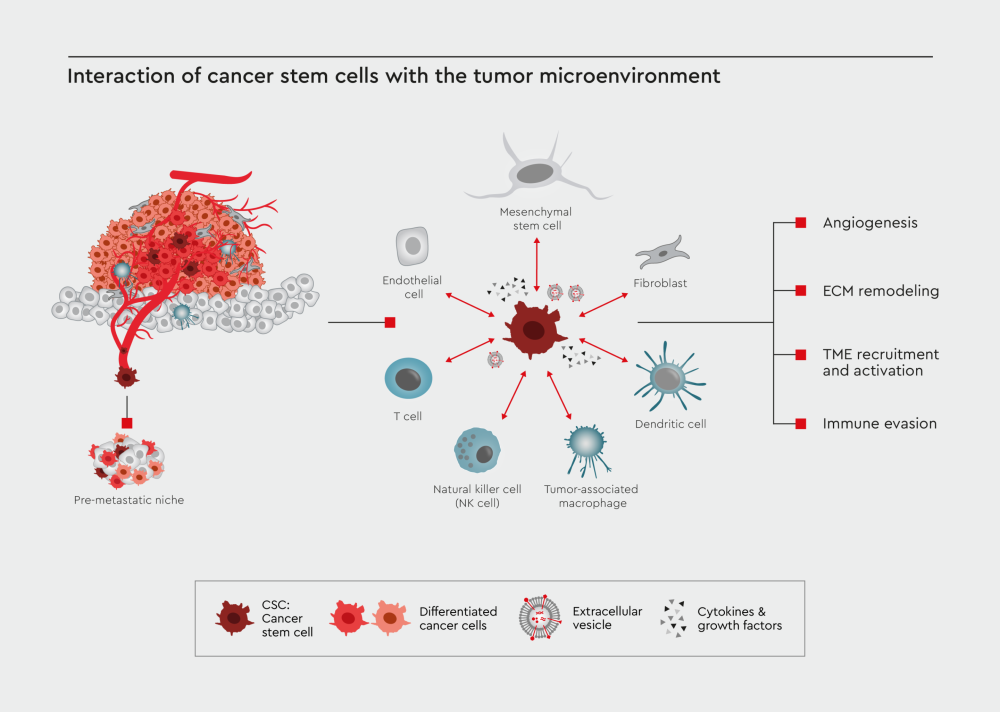

Figure 2: Workflow for establishing 3D organoids consisting of cancer and non-cancer cell types in the TME. Cancer cell culture: In our cancer media application pages, we describe a range of methodologies for obtaining primary cancer cell lines. The Primary Cancer Culture System (PCCS) is designed to help you obtain pure CSC cultures. Non-cancer cells: In the next section, we describe how you can obtain tumor-associated macrophages (TAMs) from biopsy specimens using one of our media. ECM components: Several ECM components are compatible with our cancer media. Organoid formation: Our cancer cell media are suitable for cancer organoid cultures. The choice of culture medium depends on tumor grade (aggressiveness) and presence of non-cancer cell types.
The interactions between CSCs and the tumor microenvironment have an impact on the design of therapeutic strategies. Hence, all our cancer cell media facilitate the maintenance of a healthy CSC population, which not only drives tumor progression in cell culture but also preserves the original tumor traits and crosstalk with the tumor microenvironment.
Reproducibility and scalability are key to successful development of drug discovery assays. Our cancer cell media allow you to establish primary cancer cell lines and obtain frozen cell stocks. In the last section, we provide an overview of applications for building drug testing assays, from high-throughput screening to sophisticated secondary assays.
Isolating tumor-associated macrophages – a case study
Tumor-associated macrophages (TAMs) are an important component of the cancer microenvironment. After being educated by cancer cells, TAMs adopt an anti-inflammatory, pro-tumorigenic, and pro-metastatic M2-like phenotype, thereby promoting tumor progression. TAMs can be isolated from primary tissue samples of solid tumors as non-proliferating adherent cells and can be cultured for at least two weeks.
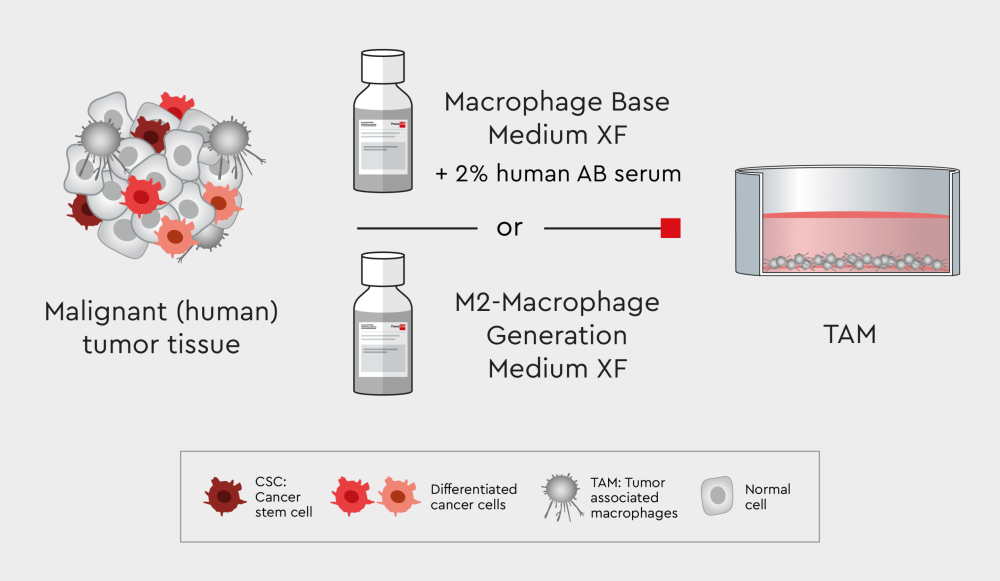

Figure 3: Workflow for isolating TAMs using our xeno- and serum-free Macrophage Media XF.
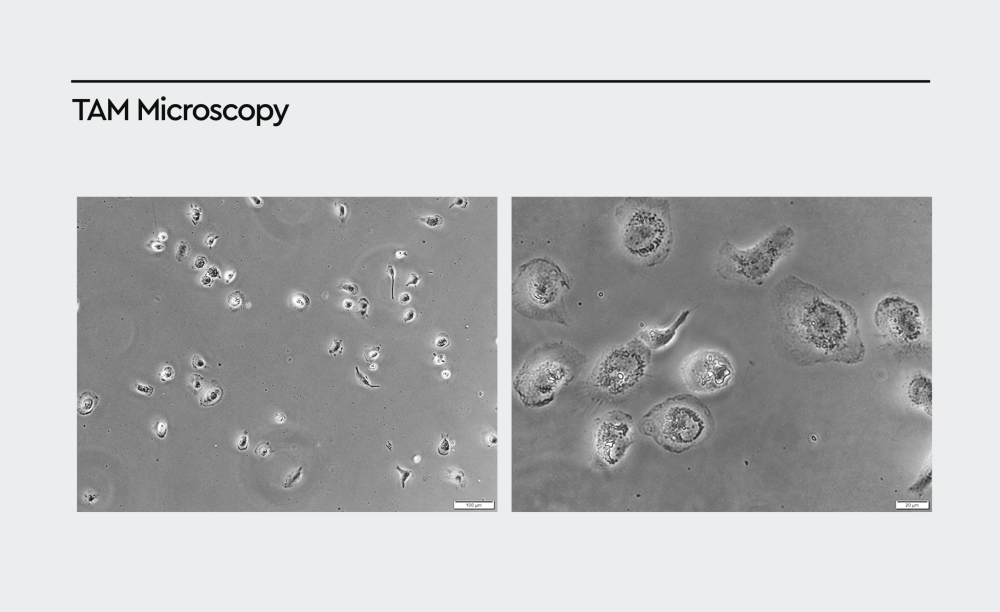

Figure 4: Phase-contrast images of TAMs isolated from primary human tumor tissue. Primary TAMs show a characteristic “fried-egg” appearance resembling M2-like macrophages. Images were taken four days after isolation (magnification: left 100x, right 400x).
Constructing and simulating the in vivo tumor microenvironment in in vitro cultures is challenging. The intended degree of similarity between in vitro conditions and the in vivo tumor microenvironment may depend on your research question or application (e.g. drug testing assays for high-throughput screening). We describe a range of applications for development of drug discovery assays on the following websites — check them out or contact our technical service team for further questions.
We are here to help you identify the right media for your cancer research.
Have a look at other Research Areas
PromoCell uses HubSpot to provide LiveChat support. This feature is currently blocked due to your cookie preferences.
Accept cookies to enable LiveChat