Innovating mouse research such as PDX mouse models is key to improving cancer therapeutics. In translating scientific findings from mice to men, multiple techniques of collecting biological data are necessary. This article will explain how.
From trying to understand the mechanisms of cancer stem cells to establishing personalized cancer therapies, in vivo, in vitro, and ex vivo cell culture techniques play a significant role in revealing crucial information to help the oncology industry progress, and robust cell culture methods are particularly necessary for the study of cancer biology. Patient-derived xenograft (PDX) mouse and rat models are heavily relied upon in cancer drug development studies to provide valuable information about the safety and efficacy of drug candidates when it comes to developing cancer therapies.1 However, the PDX mouse model is cost-intensive, tedious, and time-consuming for high-throughput drug screenings and restricted to in vivo use. On top of this, many drug prospects still fail in the clinical trial stage despite showing initial promise. Thus, amplifying PDX studies with ex vivo cell culture methods will hopefully help to increase the relevancy of data and efficacy of laboratory workflows compared to relying on in vivo studies.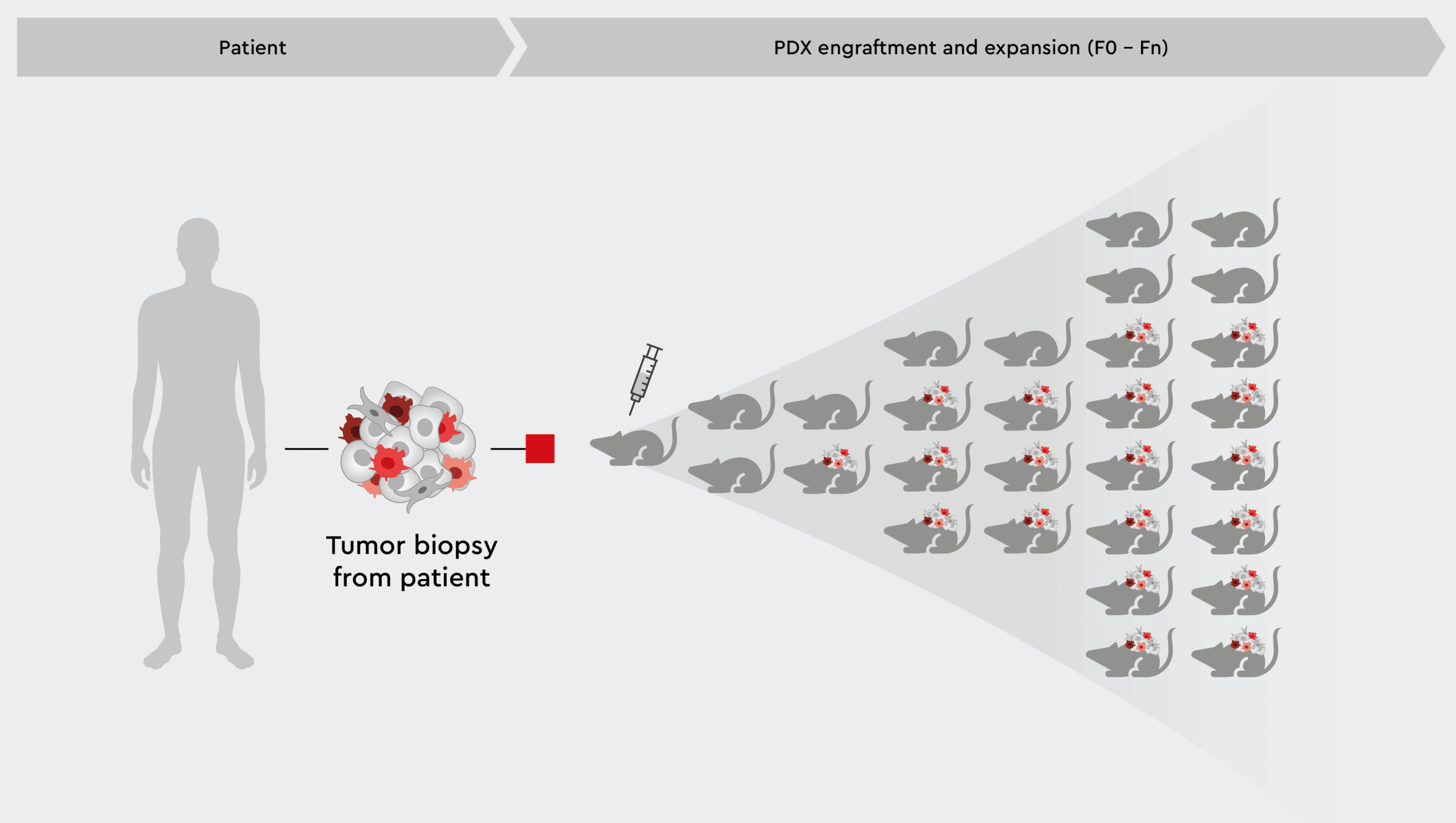
Fig. 1: Patient-derived xenograft (PDX) mouse and rat models
Schematic illustration of PDX engraftment and expansion in mouse and rat models.
Advantages of the PDX mouse model
PDX are generated through the implantation of patient tumor samples into animal models – typically mouse or rat – that are immunosuppressed and repeatedly transplanted to encourage cancer growth and progression.2 Original patient tumor characteristics are maintained in PDX mouse models such as:- cell structure
- gene expression
- metastatic potential
- genetic stability

Fig. 2: Advantages and limitations of the PDX mouse model
Overview of possibilities and restrictions of the PDX mouse model.
Limitations of the PDX mouse model
The use of PDX murine models can be expensive in terms of both money and resources. It is also considered to be rather time-consuming and with variable degrees of success in translatability for patients.3 For example, tumors could fail to grow and metastasize and lose some of the original qualities of the original transplanted tumor samples, ultimately destroying the translatability of the study.2 Tumors are highly heterogenous and rely solely on samples to engraft and transplant into mice and thus may not accurately represent human tumor morphology, meaning that they lack important data relating to how that cancer may behave and interact in a biological system – ultimately affecting the translatability value of PDX models.2 Genetic changes can occur every time cancer is transplanted into a new host, compromising the genetic traits of the original tumor.2 Moreover, the mice that are used in PDX models cannot truly reflect what would happen in the case of the human immune system as they have a compromised immune system.2 Furthermore, the tumor microenvironment in PDX mouse is not clearly understood and evolves over time, potentially leading to different biological responses between mice and humans when studying cancer drugs.3 With variable success in creating effective PDX mouse models and bearing in mind that drug candidates in clinical trials are tested solely on PDX data, these disadvantages must be addressed. Ex vivo studies using PDX material for cell culture ensure that the original tumor biology is conserved, findings remain translatable and provide a more cost-effective and less timely way to extract physiologically relevant data, creating more opportunities for more intensive research into drug molecules in clinical trials.1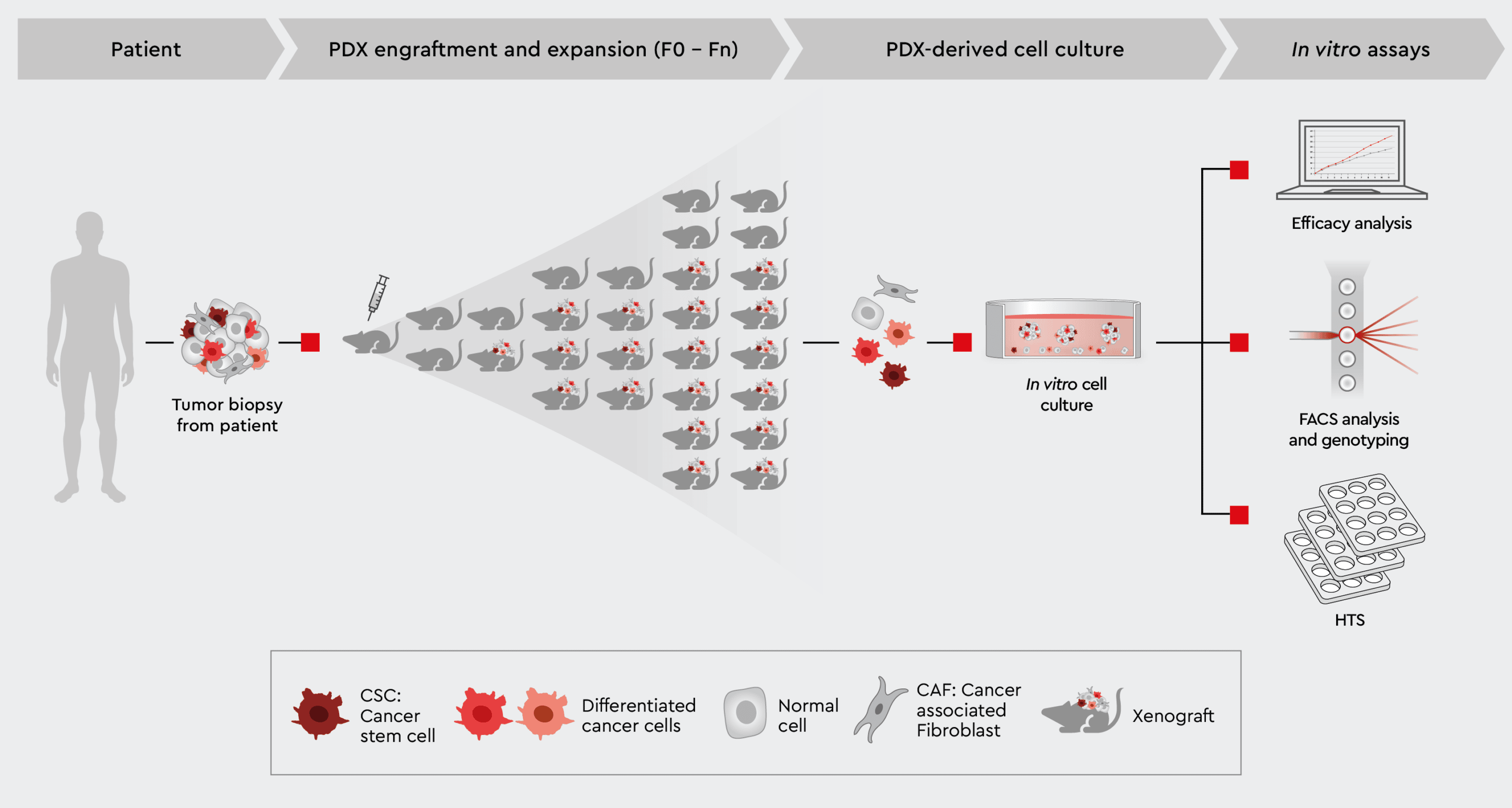
Fig. 3: Schematic illustration of the PDX model
Overview of patient-derived xenograft (PDX) model and subsequent ex vivo assays.
Innovating solutions to PDX with ex vivo cell culture
To improve the comparability of specific data from PDX models and provide answers to certain questions that can arise during animal model experiments, ex vivo cell culture studies are extremely valuable.1 For example, after observing drug failure in a PDX model, ex vivo cell culture provides a platform to isolate areas of improvement for the molecule before attempting a second round of PDX studies.1 Our experts have developed a cancer media portfolio including different functional media that allows users to establish 2D and 3D tumor models ex vivo from PDX mouse-derived tumor models. The portfolio includes media solutions for establishing primary cell lines from PDX models for testing and screening assays,4 creating master stocks of cells for later studies, and developing 3D tumor models like spheroids and organoids to address more complicated tumor biology questions.5 To make the best use of PDX mouse models, it is important to have a reliable way of expanding PDX-derived cancer cells ex vivo. Mouse cells are a common issue with conventional media when isolating cancer cells from biopsies. Cancer cells can be easily overgrown by stroma cells, and especially in PDX-derived cell cultures, the mouse cells impact the interpretation of data. This challenge can be solved by isolating primary human cancer cells from a PDX mouse and then expanding them for further assays. Our Primary Cancer Culture System6 eliminates mouse cells, and therefore only the human cells grow in the cell culture after isolation.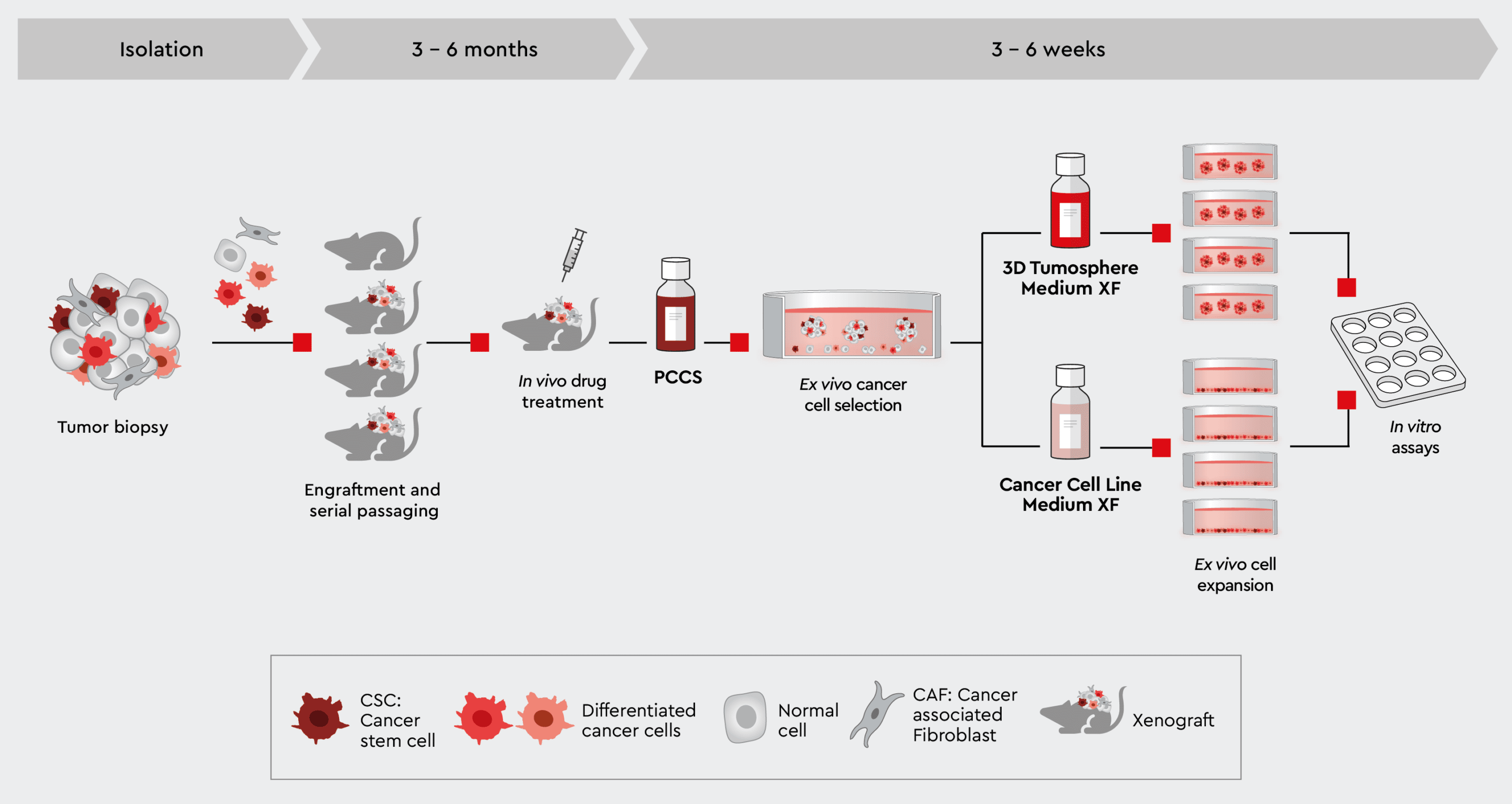
Fig. 4: PDX-derived 2D and 3D tumor models
Establish PDX-derived 2D and 3D in vitro tumor models. Expanding your PDX-derived cells in vitro in our cancer media allows you to perform high-throughput screenings, secondary assays or even organoid models while conserving the tumor cell characteristics.
References
- Making best use of your PDX mouse. (2022, September 22). PromoCell.
- Pompili, L., et al. (2016). Patient-derived xenografts: a relevant preclinical model for drug development. Journal of Experimental & Clinical Cancer Research, 35(1).
- Primary Cancer Cells: A Culture System Opens the Door to Individualized Therapy. (2017, December 11). PromoCell.
- Cancer stem cells in tumor biology. (2022, November 7). PromoCell.
- A Home for Cancer Stem Cells: 3D Tumorspheres. (2018, April 17). PromoCell.
- Primary Cancer Culture System. (2022, August 10). PromoCell.
How to make the best use of your PDX mouse
Discover how you can make the best use of your PDX mouse and how we can support you in this.
Learn more
Related pages

Application: Making best use of your PDX mouse
The PDX mouse is the working model for cancer therapies. The toolbox helps leveraging your PDX mouse project for better decision making and translation into clinic.

Application: From biopsy to assay development
Are you working with patient-derived cancer cultures? Find out, how our toolbox helps you in setting up such cancer models and build advanced assays from your biopsy.




