Air liquid interface (ALI)-cultures are useful tools that allow the study of pulmonary models using primary human airway epithelial cells such as Human Bronchial Epithelial Cells (HBEpC). Since epithelial cells provide a protective layer between two environments, this epithelial barrier can be used to study drug uptake or cytotoxicity.
Epithelial cell barrier formation can be quantitatively measured using transepithelial electrical resistance (TEER) with a voltohmmeter. To establish ALI-cultures using our Air-Liquid Interface (ALI-Airway) Medium, cells need to seed on collagen type I coated Transwell® inserts and be allowed to grow to 100% confluence in our Airway Epithelial Cell Growth Medium in a liquid-covered culture.
Seeding of cells in early passage is best, with passage 3 giving optimal results. After reaching confluence, the medium should be switched to the ALI-Airway Medium and cells in the inserts should be exposed to air (airlift). Cells will then be stimulated by the effects of airlift and the ALI-Airway Medium to develop many tight junctions to form a selective barrier on the porous membrane of the insert.
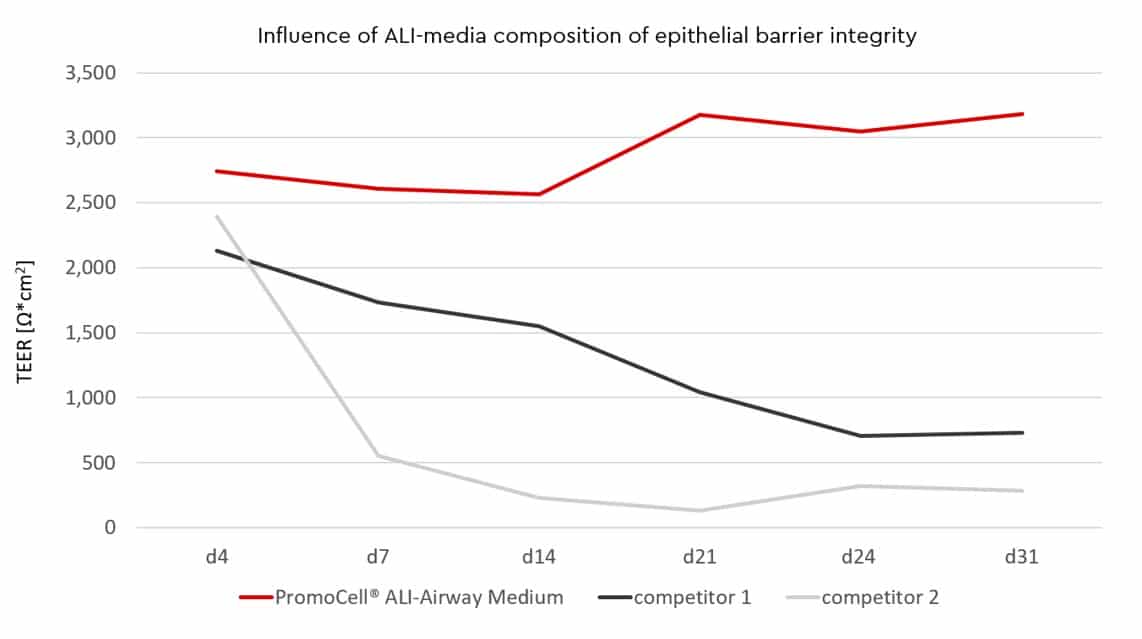
To monitor barrier formation in real time the voltohmmeter with STX2 Chopstick® electrode set can be used without damaging the cells (e.g. World Precision Instruments®). From day 3 to 4 post-airlift epithelial barrier formation is significantly better in our ALI-Airway Medium compared to competitor 1 and competitor 2, as indicated by TEER-values. The early establishment of barrier integrity makes it easy to use this pulmonary model quickly without waiting for a complete differentiation.
In the first week of ALI-culture you can achieve guaranteed TEER values > 100 Ω*cm2 with our ALI-Airway Medium. For some ALI pre-screened HBEpC donors, as shown in the graph, the values even tend to be over > 1000 Ω*cm2. TEER values > 100 Ω*cm2 have been shown to be physiological relevant in rabbit airway epithelium [1]. Other commercially available ALI-media tend to decline in their TEER-values after one week of ALI-culture. Instead of a decline of TEER-vales after the first week in ALI-culture our ALI-Airway Medium maintains the level of barrier formation.
In the second week of airlift we guarantee TEER values > 500 Ω*cm2. These TEER values are typically for HBEpC and can also be found in the literature in a range of 700 – 3.200 Ω*cm2 [2][3][4]. Even in week 3 and 4 after starting the ALI-culture barrier integrity will not decline under a threshold of > 500 Ω*cm2 when using Transwell®️ Costar®️3470 Corning®️coated with Collagen Typ I solution. Optimal barrier integrity with a high degree of viable cells (> 70% viability) will be available for at least 14 days. The choice of the right ALI-medium is therefore essential for successful ALI-cultures.
The use of minimal or complex ALI-media have a large impact on the curve progression of TEER values [5][6][7]. Other variations in TEER-values depend on the donor of the cell [8], the passage number of the cells, the right point of airlift with 100% confluent cells [9] and the temperature during TEER measurements [10]. Physiological temperature (37°C) of the ALI-culture is recommended during TEER measurements. Rapid handling during measurements can reduce heat loss. Our ALI-Airway system minimizes variations and guarantees robust and reliable TEER-values over a period of at least 14 days.
References
1. Bhat, M., et al., Regulation of tight junction permeability by calcium mediators and cell cytoskeleton in rabbit tracheal epithelium. Pharmaceutical research, 1993. 10(7): p. 991-997.
2. Lin, H., et al., Air‐liquid interface (ALI) culture of human bronchial epithelial cell monolayers as an in vitro model for airway drug transport studies. Journal of pharmaceutical sciences, 2007. 96(2): p. 341-350.
3. Oshima, T., et al., Establishment of esophageal-like non-keratinized stratified epithelium using normal human bronchial epithelial cells. American Journal of Physiology-Cell Physiology, 2011. 300(6): p. C1422-C1429.
4. Yoo, J.-W., et al., Serially passaged human nasal epithelial cell monolayer for in vitro drug transport studies. Pharmaceutical research, 2003. 20(10): p. 1690-1696.
5. Delgado-Ortega, M., et al., Newborn pig trachea cell line cultured in air-liquid interface conditions allows a partial in vitro representation of the porcine upper airway tissue. BMC cell biology, 2014. 15(1): p. 14.
6. Murgia, C., et al., Apical localization of zinc transporter ZnT4 in human airway epithelial cells and its loss in a murine model of allergic airway inflammation. Nutrients, 2011. 3(11): p. 910-928.
7. Salter, K.D., et al., Modified culture conditions enhance expression of differentiated phenotypic properties of normal rat cholangiocytes. Laboratory investigation, 2000. 80(11): p. 1775.
8. Torr, E., et al., Expression of polycomb protein BMI‐1 maintains the plasticity of basal bronchial epithelial cells. Physiological Reports, 2016. 4(16): p. e12847.
9. Forbes, B., et al., The human bronchial epithelial cell line 16HBE14o− as a model system of the airways for studying drug transport. International Journal of Pharmaceutics, 2003. 257(1): p. 161-167.
10. Srinivasan, B., et al., TEER measurement techniques for in vitro barrier model systems. Journal of laboratory automation, 2015. 20(2): p. 107-126.

