Macrophages: Key immune cells
What are macrophages?
Macrophages are specialized white blood cells that regulate the immune system and act as phagocytes. They identify and engulf harmful microorganisms, dead cells, and foreign particles through a process known as phagocytosis, serving as the first line of defense.1

What do macrophages do?
Macrophages function as key components of the innate immune system that maintain tissue homeostasis, promote wound repair, and regulate the immune responses, according to local tissue requirements. They act as sentinels, sensing and responding to changes in the microenvironment to preserve tissue integrity and balance.2,3
Key functions
- Phagocytosis and pathogen clearance: These phagocytic cells engulf and digest pathogens, dead cells, and foreign particles. They utilize pattern recognition receptors (PRRs), such as Toll-like receptors and scavenger receptors, to identify and internalize these threats through phagocytosis.4
- Immune response initiation: As first responders, they secrete pro-inflammatory cytokines and chemokines that recruit other immune cells, such as neutrophils and dendritic cells, to the site of infection. This process helps orchestrate a coordinated immune response.5
- Antigen presentation: As antigen-presenting cells (APCs) macrophages process and present antigens to T cells via major histocompatibility complex (MHC) class II molecules, bridging the innate and adaptive branches of the immune system. This activation of T cells is crucial for a robust immune response.6
- Tissue homeostasis and repair: They play a vital role in tissue development, repair, and maintenance. Macrophages also clear cellular debris and promote wound healing, which is essential for tissue regeneration.7,8
- Regulation of inflammation: These cells can exhibit both pro-inflammatory and anti-inflammatory properties, depending on their activation state. This adaptability allows them to regulate immune responses and prevent excessive inflammation that can lead to tissue damage.9
- Metabolic functions: They are involved in various metabolic processes, including iron homeostasis and regulation of reactive oxygen species (ROS). These functions are crucial for maintaining cellular health and preventing disease.10
- Disease modulation: Macrophages are implicated in various diseases, including cancer, metabolic disorders, and autoimmune conditions. Their ability to influence the tumor microenvironment makes them potential targets for therapeutic interventions.10
What are the different types of macrophages?
Macrophages exhibit phenotypic diversity based on their origin and activation state.
What are tissue resident macrophages?
Tissue resident macrophages are specialized immune cell populations found in specific tissues and organs. The table below provides examples of their different types and key functions.
| Tissue/Organ | Specialized macrophage type | Key functions |
|---|---|---|
| Placenta | Hofbauer cells (HBCs) | Placental macrophages located in the chorionic villi during gestation. They exhibit an M2-like phenotype and play important roles in placental development, including vasculogenesis and angiogenesis. 13 |
| Lung | Alveolar macrophages | Clear inhaled pathogens and particulate matter, contributing to lung immune defense and maintenance of respiratory homeostasis. 7 |
| Brain | Microglia | Resident macrophages of the brain that regulate neuroimmune responses, support tissue repair, and maintain central nervous system homeostasis. 12 |
| Liver | Kupffer cells | Involved in pathogen clearance, removal of debris, and maintenance of liver homeostasis. 11 |
| Adipose tissue | Adipose tissue macrophages (ATMs) | Maintain tissue homeostasis and respond to metabolic stress. These macrophages can polarize toward pro- or anti-inflammatory phenotypes depending on environmental and molecular cues. 14,15 |
Table 1: Examples of different tissue resident macrophages and their key functions.
Monocyte-derived macrophages
Derived from monocytes, these cells are part of the mononuclear phagocyte system and are distributed throughout the body. During infection or inflammation, monocyte-derived macrophages are recruited from the bloodstream to the inflamed site and differentiate into different subtypes depending on the local environment.16
- Classically activated (M1) Macrophages: Pro-inflammatory phagocytic cells involved in pathogen clearance and anti-tumor responses.17
- Alternatively activated (M2) Macrophages: Anti-inflammatory cells, promoting tissue repair and regeneration.17
- Regulatory macrophages (Mregs): Specialized suppressors that play a crucial role in modulating immune responses and promoting tolerance.18
Macrophage activation and polarization
Macrophage polarization is the process by which monocytes differentiate into distinct macrophage subtypes, such as M1 macrophages or M2 macrophages, under defined cytokine conditions (e.g., GM-CSF or M-CSF), resulting in non-activated phenotype-specific cells. Macrophage activation refers to the subsequent stimulation of polarized macrophages by microbial products or cytokines (e.g., lipopolysaccharide, interferon-γ, IL-4, or IL-13), triggering subtype-specific cytokine secretion and functional responses such as inflammation, immunomodulation, or tissue repair.
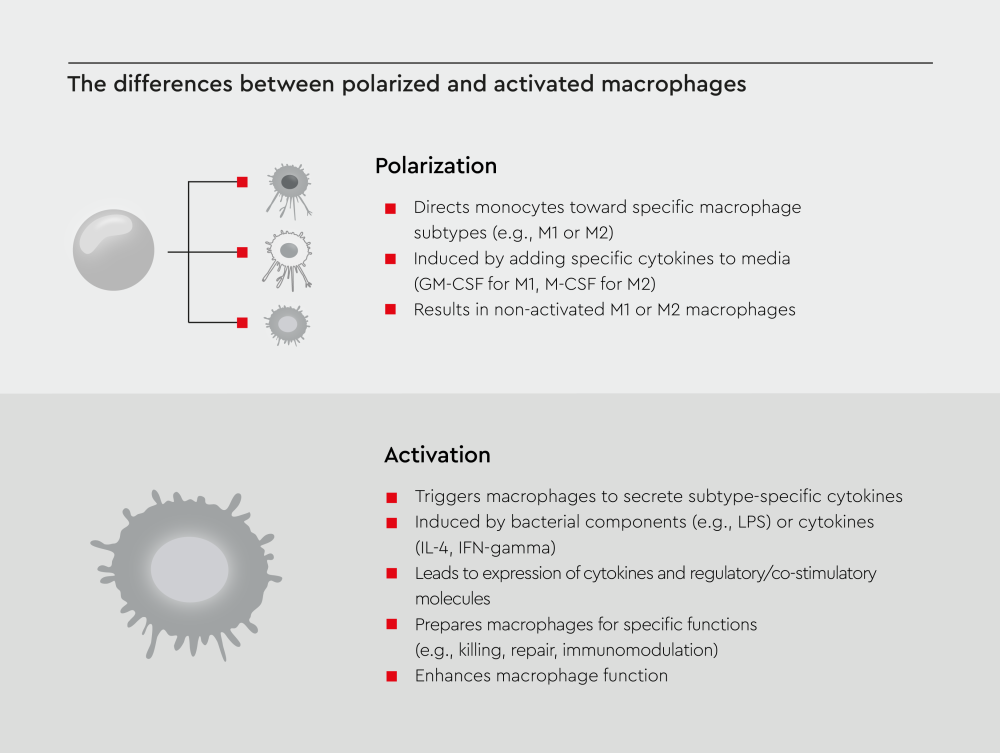

Figure 1: Schematic overview of the difference between macrophage polarization and activation.
Note: Non-activated macrophages can still function, but their activation significantly improves their performance.
What factors influence macrophage activation?
Macrophage activation is controlled by a range of signals from the cellular environment. These include immune mediators, microbial components, and metabolic conditions that shape macrophage behaviour and function. Together, these factors determine whether macrophages adopt pro-inflammatory or anti-inflammatory roles.10,19,20
The most important influencing factors include:
- Cytokines and chemokines
- Pro-inflammatory M1 activation
- Anti-inflammatory M2 activation
- Environmental conditions (pH Levels, hypoxia)
- Signaling pathways
- Metabolic pathways
- Other modulators (hormones, neurotransmitters, calcium signaling)
Macrophages in disease
These cells play multifaceted roles in disease, contributing to both protective immune responses and pathological processes:
- Cancer: Macrophages can promote or suppress tumor growth, angiogenesis, and metastasis depending on their activation state.21 M1 subtypes eliminate tumor cells through phagocytosis and cytokine release, whereas M2 subtypes promote angiogenesis and immune evasion by secreting growth factors and suppressing anti-tumor immune responses (e.g., tumor-associated macrophages (TAMs)).22–24
- Chronic inflammation: They perpetuate chronic inflammation by secreting cytokines, chemokines, and matrix metalloproteinases (MMPs), exacerbating tissue damage and promoting angiogenesis.3,25
- Autoimmune diseases: Dysregulated macrophage activity contributes to conditions such as rheumatoid arthritis and lupus.17,26
- Infections: Macrophages use pattern recognition receptors (PRRs) to detect pathogens and produce cytokines to recruit other immune cells. Although they can control viral replication and modulate inflammation, some pathogens evade digestion, leading to chronic infections.17
- Neurodegenerative diseases: Macrophages play a dual role in neurodegenerative diseases. They help clear pathological protein aggregates through phagocytosis; however, excessive uptake can impair their function, leading to neuroinflammation and neurodegeneration.27,28
- Metabolic disorders: Adipose tissue induces M1 polarization, and M1 subtypes release pro-inflammatory factors (such as TNF-α, IL-6, and IL-1β) that promote further M1 infiltration and induce insulin resistance.21,29
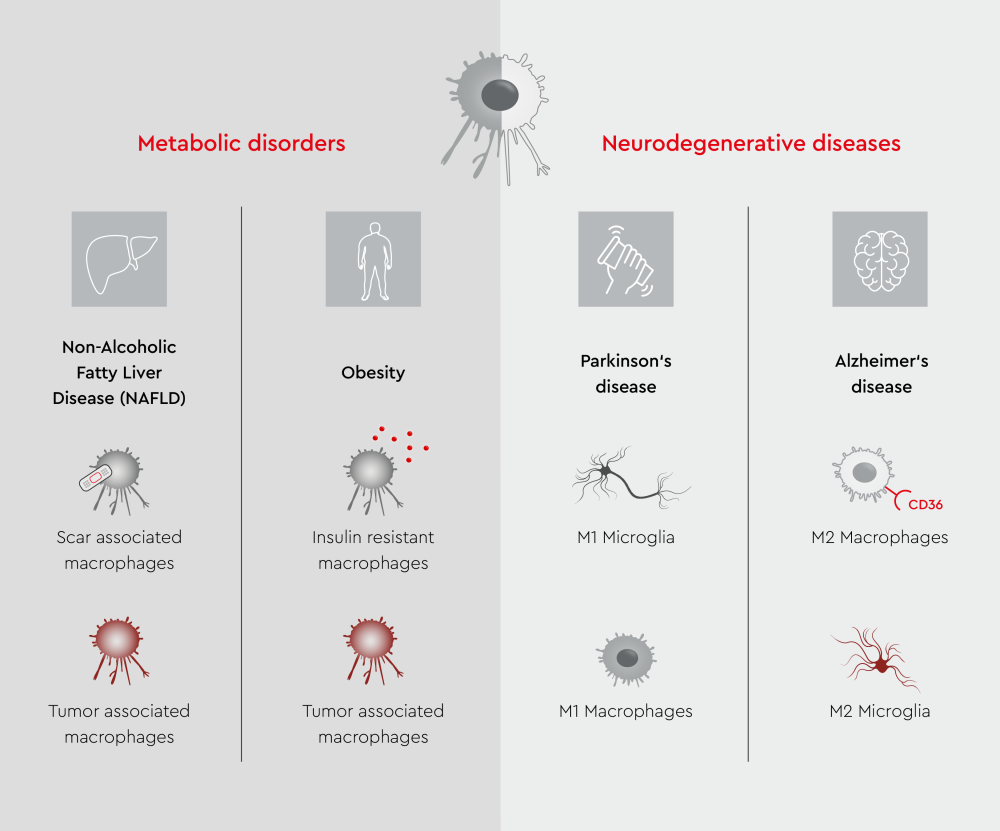

Figure 2: Overview of macrophage and microglial functional states in metabolic and neurodegenerative diseases.
Common applications of macrophages in biomedical research
Macrophages represent a frontier in biomedical research owing to their versatility. Although they serve as our body’s first line of defense against pathogens, their ability to promote both tissue homeostasis and disease pathology makes them an attractive therapeutic target. In addition, their plasticity and tissue-specific functions offer opportunities for targeted therapeutic interventions. Scientists are exploring macrophage-based cell therapies for cancer immunotherapy, tissue regeneration, and inflammatory disease treatment.30
FAQs about macrophages
FAQs about macrophages
1. How do tissue-resident macrophages differ from monocyte-derived macrophages?
Tissue-resident macrophages31-34- Originate from embryonic progenitors, including yolk sac-derived macrophages and fetal monocytes
- Seed tissues during early development and persist into adulthood through self-renewal
- Are long-lived cells that maintain stable populations under steady-state conditions
- Exhibit functional heterogeneity due to tissue-specific microenvironments
- Play critical roles in tissue development, homeostasis, and immune surveillance
Monocyte-derived macrophages31,33,35
- Arise from hematopoietic stem cells in the bone marrow, which produce circulating monocytes
- Migrate into tissues during inflammation or tissue repair and differentiate into macrophages
- Are typically short-lived and rely on continuous replenishment from circulating monocytes
- Can expand rapidly during inflammatory responses or tissue damage
- Primarily involved in inflammatory responses, pathogen clearance, and tissue repair
While monocyte-derived macrophages may eventually resemble tissue-resident macrophages in phenotype and function, they remain distinct in origin.31-34
The development and maintenance of these two macrophage populations are influenced by different factors, contributing to their unique roles in tissue homeostasis and immune responses.7,31-34
2. What are the key roles of macrophages in the immune system?
They are involved in various processes of innate and adaptive immunity, including antigen presentation, phagocytosis, cytokine production, tissue repair, and lymphangiogenesis.1,36,373. How do macrophages differ from other immune cells?
These cells are professional phagocytes that primarily function in innate immunity, maintaining inflammatory responses in tissues and clearing pathogens and dead cells.38,39 Unlike dendritic cells (DCs), which migrate to lymph nodes to present antigens, macrophages generally operate locally. They express specific markers such as CD14, CD163, C5R1, and FcγR1A, distinguishing them from other immune cells.38,394. What are the consequences of macrophage dysfunction?
Macrophage dysfunction is implicated in chronic inflammation and poor wound healing. This dysfunction can undermine the efficacy of both innate and adaptive immune systems, potentially leading to age-associated immune decline.40,41 Macrophage dysfunction can also lead to failed resolution of inflammation and excessive scarring.425. How do macrophages interact with other immune cells?
Macrophages communicate with other immune cells by secreting cytokines and chemokines, which influence the behavior and activation of nearby immune cells.43,44 This cytokine/chemokine signaling allows them to orchestrate immune responses and inflammation. For example, they can stimulate T cell responses by presenting antigens and providing co-stimulatory signals.43,446. What are the differences between M1 and M2 macrophages?
M1 subtypes, also known as classically activated macrophages, are pro-inflammatory and play a crucial role in host defense against pathogens. They produce high levels of pro-inflammatory cytokines and ROS, exhibiting a cytotoxic phenotype.45,46 In contrast, M2 subtypes, also referred to as alternatively activated macrophages, are anti-inflammatory and participate in wound repair, angiogenesis, and tissue remodeling.45,467. How do macrophages contribute to chronic inflammation?
These cells contribute to chronic inflammation by secreting inflammatory cytokines and chemokines and activating transcription factors. This process is involved in the pathogenesis of conditions like chronic obstructive pulmonary disease and idiopathic pulmonary fibrosis..258. How can macrophages be targeted for drug delivery?
Nanocarriers and microcarriers are effective tools for targeting macrophages..47 These carriers can cross membrane barriers and release drugs at infection sites. Specific receptors on the cells can be targeted to improve drug delivery efficiency. For instance, muramyl tripeptide conjugation to nanoparticles can improve their loading into macrophages..489. How do macrophages influence the adaptive immune response?
Macrophages serve as important antigen-presenting cells that can activate naive T lymphocytes, forming a bridge between innate and adaptive immunity..49 They acquire and present antigens to T cells, initiating the adaptive immune response that is essential for the clearance of pathogens.10. What are the hallmark properties of healthy macrophages?
Healthy macrophages possess three hallmark properties that are crucial for their effector and sentinel functions in the immune system.
- Response specificity: They can recognize and respond to specific pathogens or threats, allowing them to mount appropriate immune responses..4
- Context dependence: They adapt their functions based on the microenvironmental cues they receive in different tissues. This property enables them to exhibit unique features and orchestrate tissue-specific responses..50
- Stimulus memory: They can retain information about previous stimuli, which influences their future responses.
11. What role do macrophages play in tumor progression?
Macrophages infiltrating tumor tissues are known as tumor-associated macrophages (TAMs). These specialized immune cells play crucial roles in tumor growth, angiogenesis, immune regulation, metastasis, and chemoresistance..51Although many TAMs exhibit an M2 phenotype that promotes tumor growth, the M1 subtype of TAMs serves as a critical mechanism for tumor elimination..51 Repolarizing M2 TAMs to the M1 phenotype may also represent a novel and effective anti-cancer strategy..51
12. How do macrophages interact with the vascular system?
Macrophages interact with the vascular system through various mechanisms, including cytokine production, receptor engagement, and gap junction communication..52 They play a crucial role in regulating vascular inflammation, blood-brain barrier integrity, and tissue homeostasis.References
Expand
- Hirayama, D., Iida, T., & Nakase, H. (2017). The phagocytic function of macrophage-enforcing innate immunity and tissue homeostasis. International Journal of Molecular Sciences, 19(1), 92. doi:10.3390/ijms19010092
- Mosser, D. M., Hamidzadeh, K., & Goncalves, R. (2021). Macrophages and the maintenance of homeostasis. Cellular & Molecular Immunology, 18(3), 579–587. doi:10.1038/s41423-020-00541-3
- Wynn, T. A., Chawla, A., & Pollard, J. W. (2013). Macrophage biology in development, homeostasis and disease. Nature, 496(7446), 445–455. doi:10.1038/nature12034
- Sheu, K. M., & Hoffmann, A. (2022). Functional hallmarks of healthy macrophage responses: their regulatory basis and disease relevance. Annual Review of Immunology, 40(1), 295–321. doi:10.1146/annurev-immunol-101320-031555
- Nobs, S. P., & Kopf, M. (2021). Tissue‑resident macrophages: guardians of organ homeostasis. Trends in Immunology, 42(6), 495–507. doi:10.1016/j.it.2021.04.007
- Arango Duque, G., & Descoteaux, A. (2014). Macrophage cytokines: involvement in immunity and infectious diseases. Frontiers in Immunology, 5, 491. doi:10.3389/fimmu.2014.00491
- Park, M. D., Silvin, A., Ginhoux, F., & Merad, M. (2022). Macrophages in health and disease. Cell, 185(23), 4259–4279. doi:10.1016/j.cell.2022.10.007
- Wculek, S. K., Dunphy, G., Heras‑Murillo, I., Mastrangelo, A., & Sancho, D. (2022). Metabolism of tissue macrophages in homeostasis and pathology. Cellular & Molecular Immunology, 19(3), 384–408. doi:10.1038/s41423-021-00791-9
- Zhang, C., Yang, M., & Ericsson, A. C. (2021). Function of macrophages in disease: current understanding on molecular mechanisms. Frontiers in Immunology, 12, 620510. doi:10.3389/fimmu.2021.620510
- Chen, S., Saeed, A. F. U. H., Liu, Q., et al. (2023). Macrophages in immunoregulation and therapeutics. Signal Transduction and Targeted Therapy, 8(1), 207. doi:10.1038/s41392-023-01452-1
- Heymann, F., & Tacke, F. (2016). Immunology in the liver — from homeostasis to disease. Nature Reviews Gastroenterology & Hepatology, 13(2), 88–110. doi:10.1038/nrgastro.2015.200
- Hu, X., Leak, R. K., Shi, Y., et al. (2015). Microglial and macrophage polarization—new prospects for brain repair. Nature Reviews Neurology, 11(1), 56–64. doi:10.1038/nrneurol.2014.207
- Reyes, L., & Golos, T. G. (2018). Hofbauer cells: their role in healthy and complicated pregnancy. Frontiers in Immunology, 9, 2628. doi:10.3389/fimmu.2018.02628
- Silva, H. M., Báfica, A., Rodrigues‑Luiz, G. F., et al. (2019). Vasculature‑associated fat macrophages readily adapt to inflammatory and metabolic challenges. Journal of Experimental Medicine, 216(4), 786–806. doi:10.1084/jem.20181049
- Pan, D., Li, G., Jiang, C., Hu, J., & Hu, X. (2023). Regulatory mechanisms of macrophage polarization in adipose tissue. Frontiers in Immunology, 14, 1149366. doi:10.3389/fimmu.2023.1149366
- Hume, D. A., Irvine, K. M., & Pridans, C. (2019). The mononuclear phagocyte system: the relationship between monocytes and macrophages. Trends in Immunology, 40(2), 98–112. doi:10.1016/j.it.2018.11.007
- Lendeckel, U., Venz, S., & Wolke, C. (2022). Macrophages: shapes and functions. ChemTexts, 8(2), 12. doi:10.1007/s40828-022-00163-4
- Hutchinson, J. A., Riquelme, P., Geissler, E. K., & Fändrich, F. (2010). Human regulatory macrophages. In C. M. Cuturi & I. Anegon (Eds.), Suppression and Regulation of Immune Responses (Vol. 677, pp. 181–192). Humana Press. doi:10.1007/978-1-60761-869-0_13
- Murray, P. J., Allen, J. E., Biswas, S. K., et al. (2014). Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity, 41(1), 14–20. doi:10.1016/j.immuni.2014.06.008
- Orecchioni, M., Ghosheh, Y., Pramod, A. B., & Ley, K. (2019). Macrophage polarization: different gene signatures in M1(LPS⁺) vs. classically and M2(LPS⁻) vs. alternatively activated macrophages. Frontiers in Immunology, 10, 1084. doi:10.3389/fimmu.2019.01084
- Guan, F., Wang, R., Yi, Z., et al. (2025). Tissue macrophages: origin, heterogeneity, biological functions, diseases and therapeutic targets. Signal Transduction and Targeted Therapy, 10(1), 93. doi:10.1038/s41392-025-02124-y
- Qian, B. Z., & Pollard, J. W. (2010). Macrophage diversity enhances tumor progression and metastasis. Cell, 141(1), 39–51. doi:10.1016/j.cell.2010.03.014
- Xu, Y., Wang, X., Liu, L., Wang, J., Wu, J., & Sun, C. (2022). Role of macrophages in tumor progression and therapy (Review). International Journal of Oncology, 60(5), 57. doi:10.3892/ijo.2022.5347
- Zhou, L., Zhao, T., Zhang, R., Chen, C., & Li, J. (2024). New insights into the role of macrophages in cancer immunotherapy. Frontiers in Immunology, 15, 1381225. doi:10.3389/fimmu.2024.1381225
- Lee, J. W., Chun, W., Lee, H. J., et al. (2021). The role of macrophages in the development of acute and chronic inflammatory lung diseases. Cells, 10(4), 897. doi:10.3390/cells10040897
- Weyand, C. M., & Goronzy, J. J. (2021). The immunology of rheumatoid arthritis. Nature Immunology, 22(1), 10–18. doi:10.1038/s41590-020-00816-x
- Gao, C., Jiang, J., Tan, Y., & Chen, S. (2023). Microglia in neurodegenerative diseases: mechanism and potential therapeutic targets. Signal Transduction and Targeted Therapy, 8(1), 359. doi:10.1038/s41392-023-01588-0
- Silvin, A., Qian, J., & Ginhoux, F. (2023). Brain macrophage development, diversity and dysregulation in health and disease. Cellular & Molecular Immunology, 20(11), 1277–1289. doi:10.1038/s41423-023-01053-6
- Caratti, G., Stifel, U., Caratti, B., et al. (2023). Glucocorticoid activation of anti‑inflammatory macrophages protects against insulin resistance. Nature Communications, 14(1), 2271. doi:10.1038/s41467-023-37831-z
- Na, Y. R., Kim, S. W., & Seok, S. H. (2023). A new era of macrophage-based cell therapy. Experimental & Molecular Medicine, 55(9), 1945–1954. doi:10.1038/s12276-023-01068-z
- Italiani, P., & Boraschi, D. (2014). From monocytes to M1/M2 macrophages: phenotypical vs. functional differentiation. Frontiers in Immunology, 5, 514. doi:10.3389/fimmu.2014.00514
- Wu, Y., & Hirschi, K. K. (2021). Tissue‑resident macrophage development and function. Frontiers in Cell and Developmental Biology, 8, 617879. doi:10.3389/fcell.2020.617879
- Epelman, S., Lavine, K. J., & Randolph, G. J. (2014). Origin and functions of tissue macrophages. Immunity, 41(1), 21–35. doi:10.1016/j.immuni.2014.06.013
- Hoeffel, G., & Ginhoux, F. (2018). Fetal monocytes and the origins of tissue‑resident macrophages. Cellular Immunology, 330, 5–15. doi:10.1016/j.cellimm.2018.01.001
- Sanin, D. E., Ge, Y., Marinković, E., et al. (2022). A common framework of monocyte‑derived macrophage activation. Science Immunology, 7(70), eabl7482. doi:10.1126/sciimmunol.abl7482
- Yadav, S., Priya, A., Borade, D. R., & Agrawal‑Rajput, R. (2023). Macrophage subsets and their role: co‑relation with colony‑stimulating factor‑1 receptor and clinical relevance. Immunological Research, 71(2), 130–152. doi:10.1007/s12026-022-09330-8
- Fu, Y. L., & Harrison, R. E. (2021). Microbial phagocytic receptors and their potential involvement in cytokine induction in macrophages. Frontiers in Immunology, 12, 662063. doi:10.3389/fimmu.2021.662063
