3D Cell Culture
Find suitable products, scientific resources or answers to your questions within our technical library FAQ.
Checkout using your account
This form is protected by reCAPTCHA - the Google Privacy Policy and Terms of Service apply.
Checkout as a new customer
Creating an account has many benefits:
What if one of the biggest influences on tumor progression isn’t the cancer cell itself, but the cells surrounding it? The tumor microenvironment (TME) contains various types of non-cancer cells, including cancer-associated fibroblasts (CAFs). Cancer-associated fibroblasts are stromal cells that can promote tumor growth, immune escape, and metastasis. Understanding cancer-associated fibroblasts is essential for the development of effective cancer treatments and overcoming drug resistance. Take a closer look at the biology of cancer-associated fibroblasts and how these cells provide a new angle on cancer discovery. This page breaks down the complex biology of cancer-associated fibroblasts to help you navigate cancer discovery with confidence.
Cancer-associated fibroblasts are activated stromal cells within the TME.1 They are abundant components of most solid tumors and play a role in cancer progression and therapy response. Recent technological advances have revealed substantial molecular diversity in CAFs, but understanding their functions has been challenging.2
CAFs can develop from various cell types, including:
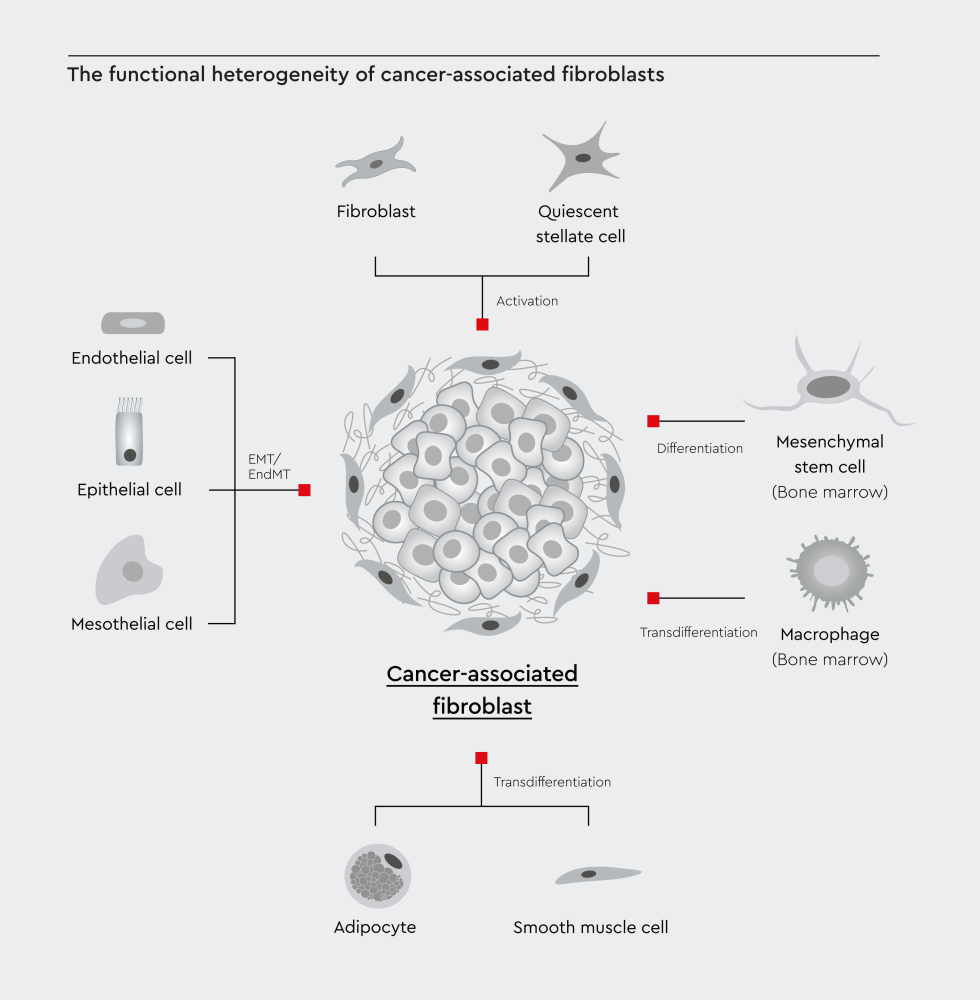

Figure 1: The functional heterogeneity of cancer-associated fibroblasts reflects the diversity of their cellular sources. Local tissue-resident fibroblasts and stellate cells can activate into CAFs. Non-fibroblast lineages, including epithelial cells, endothelial cells, adipocytes, and bone marrow-derived mesenchymal stem cells, can also convert into CAFs through cellular reprogramming. Adopted from Yang et al, 2023.6
The transformation of normal fibroblasts to activated fibroblasts is regulated by tumor growth factor-beta (TGF-β) and platelet-derived growth factor (PDGF).7,8 Mechanical stiffness in the TME also contributes to the transformation of normal fibroblasts into CAFs.9,10
Researchers identify cancer-associated fibroblasts using a combination of molecular markers. These include PDGF receptor alpha (CD140a), PDGF receptor beta (CD140b), fibroblast specific protein 1 (FSP-1/S100A4), fibroblast activation protein (FAP), and alpha-smooth muscle actin (α-SMA).11
CAFs differ from normal fibroblasts primarily in their functional roles. Normal fibroblasts regulate tissue architecture and wound healing. They produce extracellular matrix (ECM) components in a regulated manner.12 Unlike normal fibroblasts, CAFs promote tumor growth through multiple mechanisms. CAFs are activated and respond to tumor-derived signals. They promote tumor growth rather than tissue maintenance.6,13
The secretory profile of CAFs is also different from that of normal fibroblasts. CAFs release cytokines, growth factors, and ECM-modifying enzymes that aid cancer cell invasion and metastasis.14,15 They also facilitate immune evasion by creating an immunosuppressive environment.14,15
Note: Fibroblasts are not cancer cells, but their tumor-supporting functions can contribute to cancer progression. CAFs remodel the ECM to facilitate invasion, secrete pro-angiogenic factors that promote the formation of new blood vessels, and deliver pro-survival signals that aid metastasis.14,16,17
Cancer-associated fibroblasts contribute to multiple aspects of cancer biology. Their influence extends from the early stages of tumor development through metastasis.
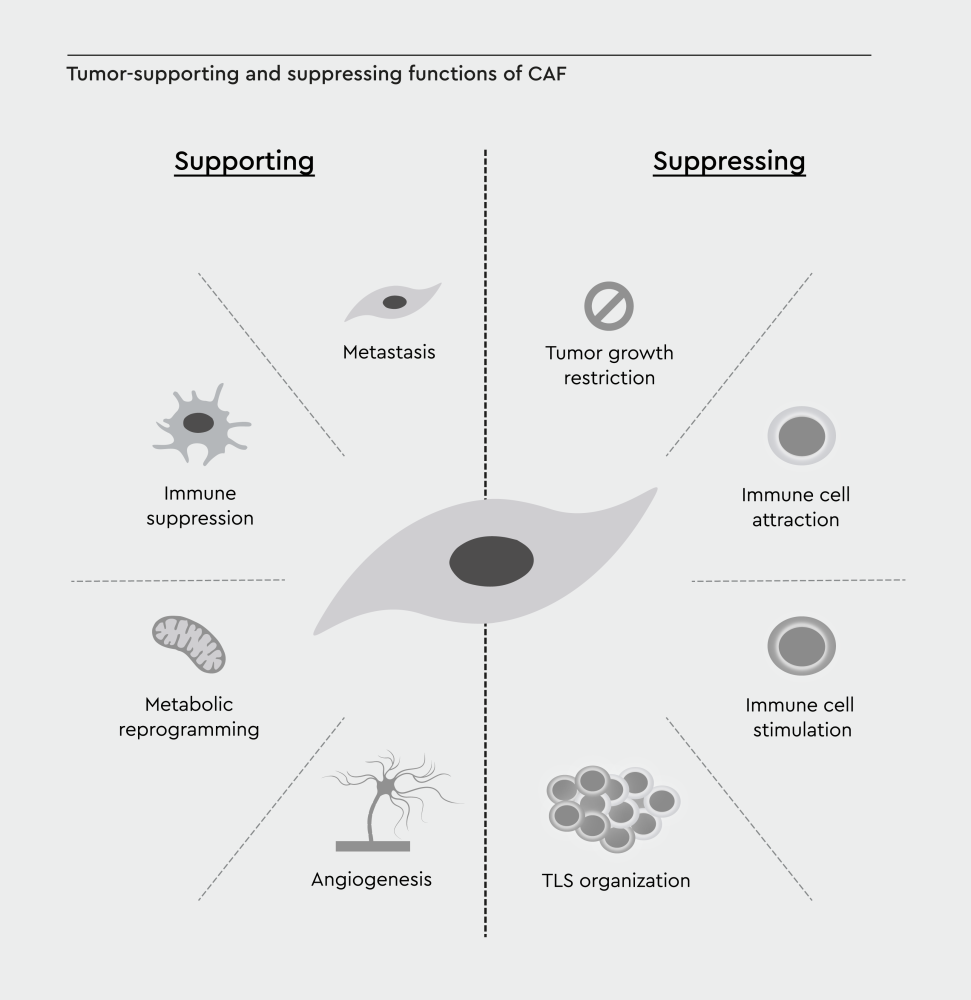

Figure 2: Hallmarks of cancer-associated fibroblasts. Tumor-supporting and tumor-suppressing functions of CAFs are connected to the hallmarks of cancer by Hanahan and Weinberg. TLS, tertiary lymphoid structure. Adopted from Cords et al., 2024.24
Cancer-associated fibroblasts are a heterogeneous population. Recent studies have identified distinct CAF subtypes, such as inflammatory CAFs (iCAFs), that have specialized functions.


Figure 3: Cancer-associated fibroblasts exist as multiple functional subtypes within the tumor microenvironment. CAF subsets include myCAFs, iCAFs, apCAFs, meCAFs, vCAFs, and tCAFs. CAF subtypes are not fixed but can interconvert in response to environmental signals.25–28 Adopted from Cords et al., 2024.24
Genetic alterations in precursor cells shape the characteristics of CAFs and can promote a shift toward specific CAF phenotypes.29 Epigenetic changes, such as DNA methylation and histone modifications, allow CAFs to maintain stable phenotypes or switch between states.29 Alterations in cellular metabolism can also influence the function of CAFs and their influence on the tumor microenvironment.29 Additionally, chemotherapy drugs can alter the TME by modifying autocrine or paracrine signaling pathways and transcription factors.30 Tumor-derived signals can also control CAF plasticity, with tumor-secreted TGF-β promoting the development of myCAF phenotypes. In contrast, IL-1 ligand secretion promotes the formation of iCAF phenotypes.31
The heterogeneity of CAFs makes the development of anti-CAF therapies challenging. Different CAF subtypes may require distinct approaches for effective targeting. In addition, some CAFs may have tumor-suppressing functions, and eliminating all CAFs could potentially worsen outcomes.32
The plasticity of cancer-associated fibroblasts also contributes to the complexity of developing therapies targeting CAFs. CAFs can shift phenotypes in response to changes in the TME. Remaining CAFs might compensate for depleted populations.33 These challenges highlight the need for better cancer models that better recapitulate the 3D environment of tumors and incorporate CAFs and other stromal cells.
For a closer look at how cancer-associated fibroblasts really behave, 3D cultures bring you much closer to the true tumor microenvironment than 2D systems ever could. Traditional 2D cultures don’t replicate ECM architecture or mechanical properties, which can influence the behavior of CAFs.34
Cell-cell and cell-matrix interactions are also better preserved in 3D cultures. In conventional 2D cultures, CAFs grow on flat surfaces with limited cell-matrix interactions.35 In 3D models, CAFs adopt more physiologically relevant functions. They demonstrate ECM remodeling, establish proper cell-cell contacts, and influence cancer cell invasion patterns that better reflect tumor behavior.35
In addition to providing a more physiologically relevant model for studying CAFs, 3D cultures also serve as a tool for testing cancer therapies. Three-dimensional models can be used to study CAF-mediated drug resistance and test anti-CAF therapies in these systems before moving to animal studies.35 Organoid models derived from patient tumors can be cultured with patient-matched CAF populations.36 These patient-specific systems enable personalized medicine approaches.
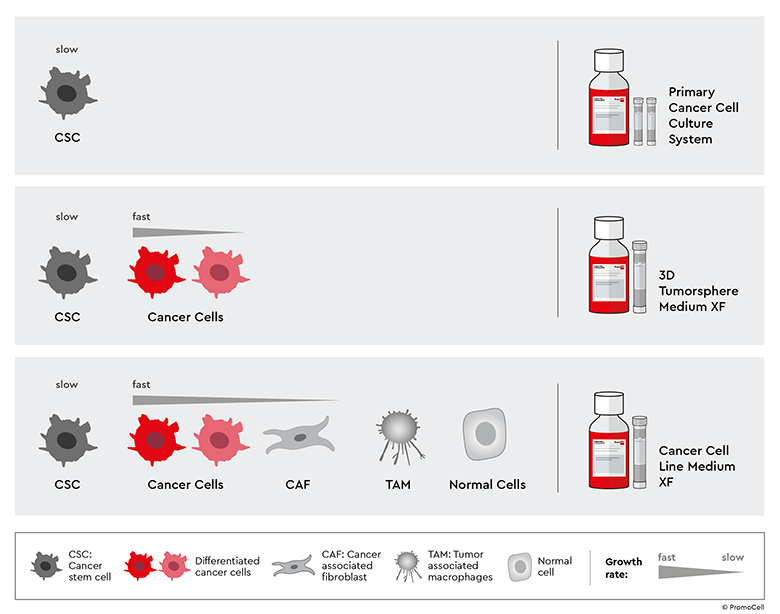
Studying cancer-associated fibroblasts requires the development of reliable and physiologically relevant cancer models. Our Primary Cancer Culture System (PCCS) enables you to establish long-term culture of cancer stem cells (CSCs) from tumor biopsies and patient-derived xenografts (PDX). This approach preserves tumor heterogeneity by allowing the isolation of CSCs, which, when co-cultured with CAFs, can be used to generate physiologically relevant 3D tumor models.
Our Cancer Cell Line Medium XF provides a standardized environment for cancer cell expansion and is ideal for their co-culture with CAFs. You can use it as a standalone solution or integrate it with PCCS and 3D Tumorosphere Medium XF. This system maintains CAF-cancer cell interactions in physiologically relevant architectures, ideal for studying ECM remodeling, invasion, and mechanisms of drug resistance.
Our cancer media toolbox also includes 3D Tumorsphere Medium XF for the isolation and long-term 3D cultivation of CSCs. It supports the enrichment and maintenance of CSCs and differentiated cancer cells, with high cell proliferation rates.

The development of 3D cancer models that preserve CAF biology may require optimization. Our technical service team is here to provide you with expert guidance for your specific research needs. We’ll help you select suitable media and culture conditions for CAF studies.
Curious how far your CAF research could go with the right tools? Fill our cancer media toolbox request form to help you push your CAF research forward.
References
Have a look at other Research Areas
PromoCell uses HubSpot to provide LiveChat support. This feature is currently blocked due to your cookie preferences.
Accept cookies to enable LiveChat