Ensure consistency across outsourced research and CRO workflows
Many biotech and pharmaceutical teams rely on CROs for critical research steps.
As workflows move between internal labs and external partners, differences in cell sources, logistics, and coordination can introduce variability. This can affect assay performance, data comparability, and study outcomes.
We help you align your outsourced research from the start. This way, your teams and CRO partners work from the same biological foundation, and your data stays consistent across sites.
Get expert input on cell models, donor availability, and CRO coordination.
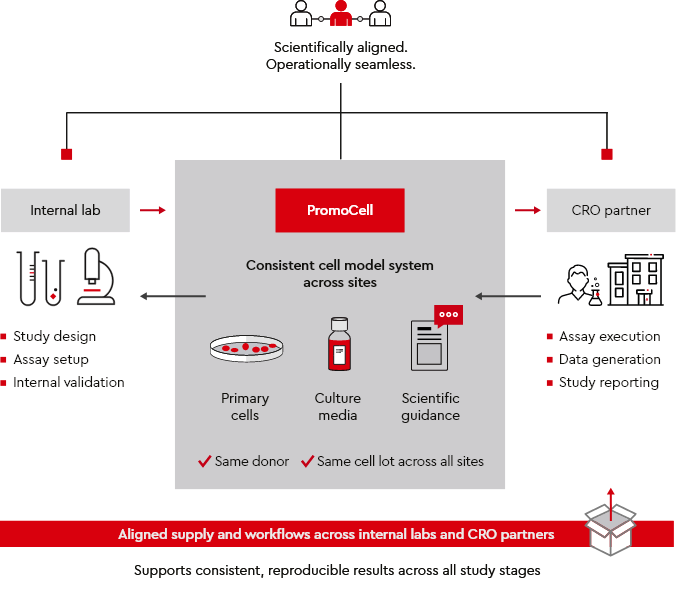

Figure 1: One system. One standard. Consistent results across all sites and study stages.
How do you ensure consistent cell models across CRO and internal labs?
We support reproducible results across internal labs and CROs through:
- Primary human cells and specialized culture media
- Consistent donor and cell lot material across sites
- Application guidance and scientific support for your team and CRO partners
This reduces variability and simplifies assay transfer across sites, while improving cross-site data comparability.
How do you maintain consistency across outsourced research projects?
Consistency matters not only across sites, but also across time. Our approach is built on continuity throughout the study duration. For long-term CRO-based studies, we support continuity through:
- Reservation of specific cell lots for the full project duration
- Consistent donor material across internal labs and CRO sites
- Access to donor-specific cell expansion solutions
- Early evaluation of materials while securing supply for the entire study duration
This improves reproducibility across time and locations. It also reduces the risk of mid-study changes in biological material.
Do your CRO-based studies require specific donor characteristics?
For studies requiring defined biological characteristics of cells, we provide:
- Access to established tissue donation networks
- Targeted donor pool selection
- Availability of diverse HLA profiles
This ensures your cell model aligns with your study requirements across CRO workflows. This is especially relevant for complex, immunology-focused, or translational research workflows.
How do you coordinate workflows between your lab and a CRO partner?
We help simplify coordination across outsourced research workflows. This includes timelines, assay transfer, and multi-site alignment.
We support your project through:
- Direct shipping to CRO locations
- Free cell lot reservations coordinated across multiple sites
- Scientific support for both your team and your CRO partner
- Flexible ordering options, including eProcurement integration
This reduces administrative effort and improves alignment across teams and timelines.
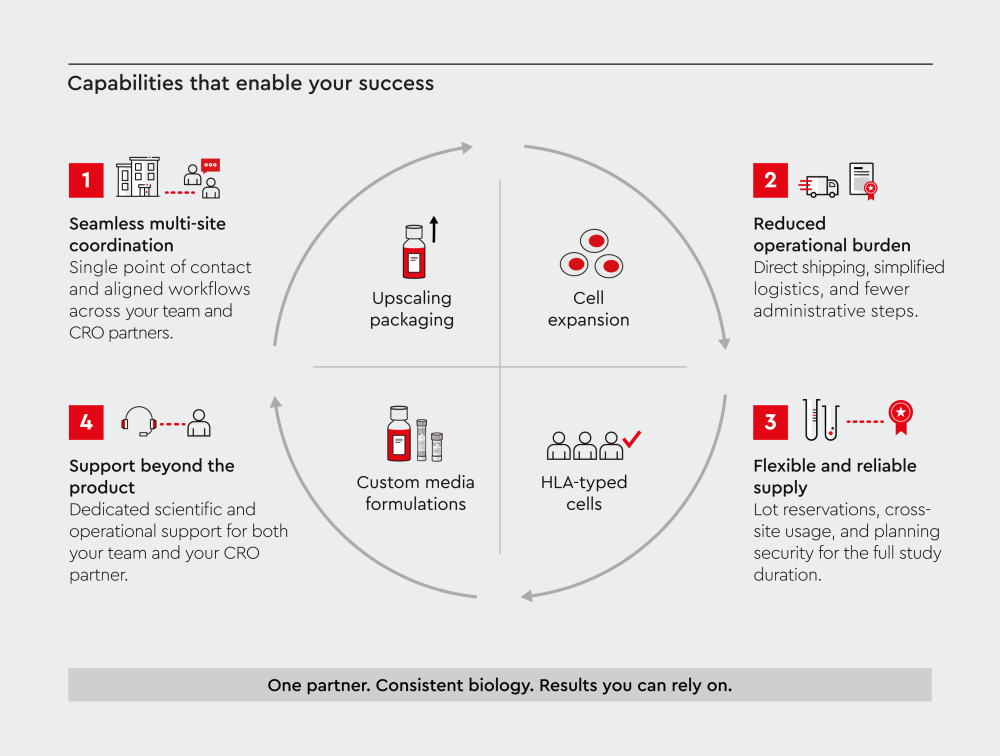

Figure 2: Coordinated CRO workflows built on consistent biology and reliable supply
What does a typical outsourced research workflow look like?
A typical CRO-based workflow includes:
- Selecting a suitable cell model system
- Reserving a donor lot for the study duration
- Shipping materials to internal labs and CRO sites
- Coordinating scientific support across teams
- Using the same biological material across all study stages
This approach supports continuity and reduces variability throughout the project lifecycle.
How do you manage requirements in outsourced research projects?
Outsourced research projects often come with requirements related to documentation, handling, or material use.
We support you by:
- Aligning your setup with project-specific needs
- Providing guidance on documentation and process requirements
- Supporting next steps where additional requirements apply
If needed, you can also contact our team directly via our dedicated email for extended right-of-use inquiries.
Some projects may also require alignment with regulatory or quality standards - for example, when research workflows move closer to translational or GMP-relevant stages. Where needed, we support these projects with our Custom GMP Services and materials, helping ensure documentation readiness, consistent sourcing, and continuity across internal labs and CRO partners.
How do you get started with a CRO-based project?
Early alignment is critical for CRO-based studies.
We help you:
- Select suitable cell models and media
- Plan donor availability and reservations
- Align workflows across internal labs and CRO partners
- Address project-specific requirements
Align your outsourced research workflow from the start Talk to our team to plan your CRO-based study, secure donor availability, and ensure consistency across all sites. Reduce risk, avoid rework, and build more reliable research workflows from day one.
