Potential of mesenchymal stem cell-derived extracellular vesicles for targeted drug delivery
Mesenchymal stem cells (MSCs) are multipotent stromal cells with immunomodulatory and regenerative properties that continuously secrete vesicles into their surrounding environment. These particles are known as extracellular vesicles (EVs). EVs carry proteins, lipids, and nucleic acids, shuttling biological information between cells throughout the body. Because of their cargo-loading capacity and ability to travel through biological fluids, EVs have emerged as an attractive platform for targeted drug delivery
EVs can be derived from a variety of cell sources, including dendritic cells, T cells, platelets, and endothelial cells. Among these, MSC-derived extracellular vesicles (MSC-EVs) have been extensively studied as potential carriers for targeted drug delivery. Ready to explore what makes MSC-EVs such a powerful platform? Read on.
What role do mesenchymal stem cell-derived extracellular vesicles play in immune response?
MSC-derived extracellular vesicles act as cell-free mediators of immunomodulation. They regulate both innate and adaptive immune responses, helping to reduce inflammation and support tissue repair.1 MSC-EVs carry a variety of bioactive molecules, including miRNA, lncRNA, cargo proteins, and lipids, which they deliver to immune cells.
MSC-EVs also show protective effects in a range of conditions, including central nervous system (CNS) disorders, myocardial infarction, liver injury, lung disease, ulcerative colitis, and kidney injury.2 This makes them an attractive tool for therapeutic use.
What’s the therapeutic potential of mesenchymal stem cell-derived extracellular vesicles?
Stem cell-derived extracellular vesicles are pushing the field toward what researchers call “cell therapy 2.0”, a cell-free approach that retains many benefits of MSC therapy while reducing some of its risks.3
Unlike whole-cell transplants (e.g., stem cell immunotherapy), EVs have a lower tendency to trigger immune rejection.4 They cannot form tumors directly. These safety advantages make stem cell-derived extracellular vesicles an attractive platform for targeted drug delivery systems.4
Exosomes, a subtype of EV, in particular, have low immunogenicity.5 Their surface proteins can be modified through genetic engineering to improve targeting to a specific target cell or target site.5 This makes exosomes highly adaptable as drug carriers.
Several preclinical and clinical studies are now exploring MSC-EVs as targeted delivery tools:
- Cancer treatments: EVs secreted by engineered MSCs have been loaded with anti-tumor agents and tested in glioblastoma models.6 MSC-EVs exploit the natural tumor-homing properties of their parent cells. Cancer cells also secrete EVs that play a role in tumor progression, a property that researchers are exploring for treatment strategies and as a potential diagnostic tool.
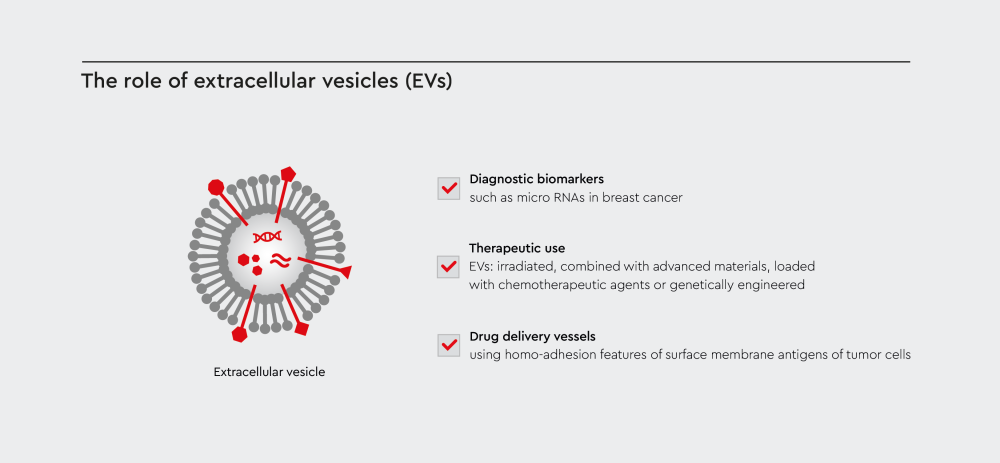

Figure 1: Roles of extracellular vesicles (EVs) in cancer diagnosis and therapy.
- Neurological disorders: MSC-EVs have been explored for crossing the blood-brain barrier to deliver neuroprotective cargo to damaged neurons, a route unavailable to many conventional drugs.7
- Lung diseases: Early-phase clinical trials have evaluated intravenous MSC-EV preparations for their anti-inflammatory effects in respiratory diseases, building on preclinical evidence of lung protection.7,8
These programs signal a move of extracellular vesicles from fundamental research to studies of their potential use as a tool for targeted drug delivery.
Scaling up MSC-EVs: Manufacturing challenges and considerations
Large-scale production of MSCs is one of the biggest challenges in the use of MSC-EVs as vehicles for drug delivery.9 A single human therapeutic dose may require EVs from hundreds of millions of MSCs.9 That demands large cell numbers, and it demands consistency.
Several factors influence MSC-EV production yield and quality, as summarized below.
| Factor | Effect on EV yield and quality | Implication for clinical use |
|---|---|---|
| MSC variability and cell source9,10 | Influence EV yield and biological quality; high-passage MSCs produce EVs with lower activity and altered cargo composition. | Well-characterized, low-passage MSCs are preferred for consistent, clinical-grade EV isolation. |
| Media choice9 | Important for scalable manufacturing and translation of MSC-derived extracellular vesicles. | Defined or xeno-free MSC culture media reduce batch-to-batch variability, improve reproducibility, and support scalable, clinical manufacturing of MSC-EVs. |
Table 1: Factors influencing MSC-EV production yield and quality.
What tools support mesenchymal stem cells scale-up for EV therapy?
Because EV-based therapies depend on large quantities of healthy, consistent MSCs, starting material quality isn’t something to optimize later. It needs to be built in from the very beginning, at the research stage, long before clinical development begins.
Consistency in MSC culture and EV isolation is what makes high yield and high reproducibility achievable. Variations in seeding density, passage number, or media formulation can influence the amount and quality of MSC-derived EVs, and establishing reproducible protocols early can prevent failures down the line.
At PromoCell, we offer a comprehensive range of mesenchymal stem cell products for both research and clinical applications. Our portfolio covers primary human MSCs from multiple tissue sources, specialized MSC culture media, and MSC differentiation media, all characterized for consistent, reproducible performance.
FAQs
References
Expand
- Wang JH, Liu XL, Sun JM, Yang JH, Xu DH, Yan SS. Role of mesenchymal stem cell derived extracellular vesicles in autoimmunity: A systematic review. World J Stem Cells. 2020;12(8):879-896. doi:10.4252/wjsc.v12.i8.879
- Dabrowska S, Andrzejewska A, Janowski M, Lukomska B. Immunomodulatory and regenerative effects of mesenchymal stem cells and extracellular vesicles: Therapeutic outlook for inflammatory and degenerative diseases. Front Immunol. 2021;11:591065. doi:10.3389/fimmu.2020.591065
- Rohde E, Pachler K, Gimona M. Manufacturing and characterization of extracellular vesicles from umbilical cord–derived mesenchymal stromal cells for clinical testing. Cytotherapy. 2019;21(6):581-592. doi:10.1016/j.jcyt.2018.12.006
- Wu X, Jiang J, Gu Z, Zhang J, Chen Y, Liu X. Mesenchymal stromal cell therapies: immunomodulatory properties and clinical progress. Stem Cell Res Ther. 2020;11(1):345. doi:10.1186/s13287-020-01855-9
- Elahi FM, Farwell DG, Nolta JA, Anderson JD. Preclinical translation of exosomes derived from mesenchymal stem/stromal cells. Stem Cells. 2020;38(1):15-21. doi:10.1002/stem.3061
- Agosti E, Antonietti S, Ius T, Fontanella MM, Zeppieri M, Panciani PP. A systematic review of mesenchymal stem cell-derived extracellular vesicles: A potential treatment for glioblastoma. Brain Sci. 2024;14(11):1058. doi:10.3390/brainsci14111058
- Shi J, Zhao YC, Niu ZF, et al. Mesenchymal stem cell-derived small extracellular vesicles in the treatment of human diseases: Progress and prospect. World J Stem Cells. 2021;13(1):49-63. doi:10.4252/wjsc.v13.i1.49
- Van Griensven M, Balmayor ER. Extracellular vesicles are key players in mesenchymal stem cells’ dual potential to regenerate and modulate the immune system. Adv Drug Deliv Rev. 2024;207:115203. doi:10.1016/j.addr.2024.115203
- Adlerz K, Patel D, Rowley J, Ng K, Ahsan T. Strategies for scalable manufacturing and translation of MSC-derived extracellular vesicles. Stem Cell Res. 2020;48:101978. doi:10.1016/j.scr.2020.101978
- Patel DB, Gray KM, Santharam Y, Lamichhane TN, Stroka KM, Jay SM. Impact of cell culture parameters on production and vascularization bioactivity of mesenchymal stem cell‐derived extracellular vesicles. Bioeng Transl Med. 2017;2(2):170-179. doi:10.1002/btm2.10065
- Doyle L, Wang M. Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells. 2019;8(7):727. doi:10.3390/cells8070727
- Wang Y, Zhu J, Ma Q, et al. Trends in mesenchymal stem cell-derived extracellular vesicles clinical trials 2014–2024: is efficacy optimal in a narrow dose range? Front Med. 2025;12:1625787. doi:10.3389/fmed.2025.1625787
- Matsuzaka Y, Yashiro R. Current strategies and therapeutic applications of mesenchymal stem cell-based drug delivery. Pharmaceuticals. 2024;17(6):707. doi:10.3390/ph17060707
- Maumus M, Rozier P, Boulestreau J, Jorgensen C, Noël D. Mesenchymal stem cell-derived extracellular vesicles: opportunities and challenges for clinical translation. Front Bioeng Biotechnol. 2020;8:997. doi:10.3389/fbioe.2020.00997
