Our Cryo-SFM Plus boasts an optimized formulation, standing out with its innovative and powerful antioxidant technology. This unique blend ensures uncompromised integrity and viability of human and animal cells during long-term storage.

Key characteristics of our Cryo-SFM Plus:
- Defined composition: With Cryo-SFM Plus, you obtain a precisely defined formulation for consistent results. No more guesswork — just reproducible outcomes every time.
- Protein-free: Ensure a standardized and controlled environment for your precious cell samples. Cryo-SFM Plus is free from proteins, eliminating unwanted variability.
- Animal component-free: Enjoy a regulatory-friendly format that gives you peace of mind. No animal-derived components means compliance without compromise.
Key benefits of our Cryo-SFM Plus:
- Innovative: Our antioxidant technology preserves viability, attachment, and growth.
- Optimal cell yield: Experience maximum cell recovery after thawing. Cryo-SFM Plus ensures that your experiments proceed smoothly, whether you’re working with primary cells, stem cells, or established cell lines.
- Minimal freeze damage: Protect your valuable cell samples from damage during freezing and thawing. Cryo-SFM Plus minimizes freeze-induced stress, preserving cell viability and integrity.
- Versatile: Cryo-SFM Plus adapts to your research needs and is suitable for various cell types — from delicate stem cells to established lines.
- Powered by patented technology: Rest assured with our patented* technology. Cryo-SFM Plus is backed by rigorous research and innovation, ensuring top-tier performance.
*Patent issued in Germany and pending approval in other countries.
Preserving cell viability and function during cryopreservation is particularly challenging for sensitive cell types, such as stem cells, cancer cells, and melanocytes — these cells often show impaired cell health after thawing. Cells frozen in Cryo-SFM Plus exhibit superior viability, attachment, and growth performance after thawing.
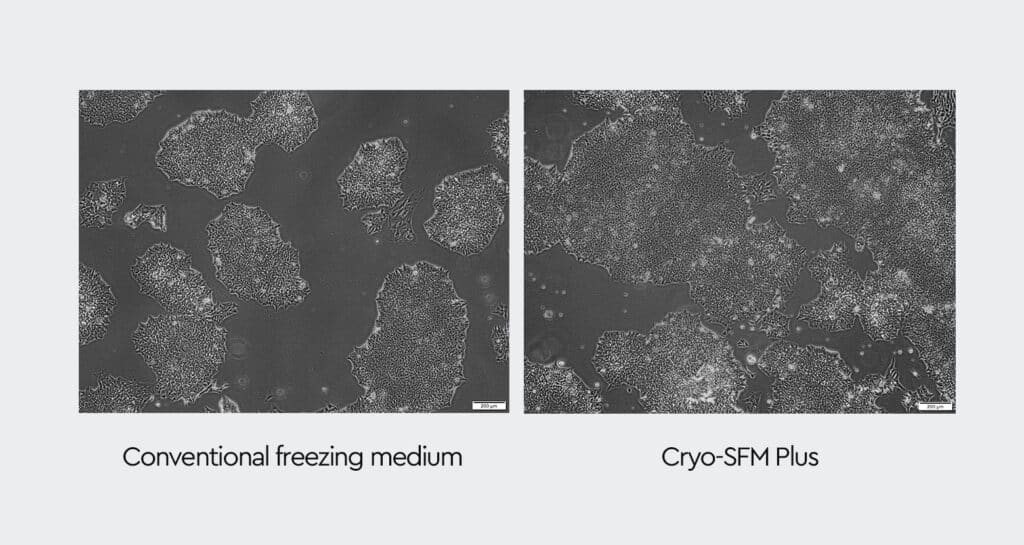
Are you tired of compromising cell functionality and performance during freezing and thawing?
Go to our product page and place your order now:

“Optimal cryopreservation is paramount for cell culture excellence. With Cryo-SFM Plus, we introduce a breakthrough solution meticulously developed to elevate cell culture standards. Through extensive testing and refining, we’ve crafted a solution that will significantly improve cell culture practices. The results speak for themselves, demonstrating enhanced post-thaw growth and reproducibility. I’m truly excited to unveil this innovation to the scientific community, inviting you to experience the transformative potential firsthand. Try our new Cryo-SFM Plus and witness its unparalleled impact on cell culture performance.”
