Cell detachment is a critical step in cell culture workflows using adherent cells. If you choose the wrong reagent or leave it on too long, you can damage your cells. Selecting the right cell detachment method directly affects cell viability and downstream results.
At a glance: Quick answers
What is cell detachment?
What is trypsinization?
What is the role of EDTA in cell detachment?
What is Accutase?
What causes cell damage during detachment?
What is cell detachment in cell culture?
Cell detachment describes the release of adherent cells from tissue culture for passaging, harvesting, or downstream use.1 It is commonly performed using enzymatic reagents such as trypsin/EDTA or gentler alternatives, depending on the cell type and experimental needs.1,2
Cell detachment becomes necessary whenever you need to:
- Subculture (passage) cells before they reach confluence
- Harvest a defined number of cells for an experiment
- Transfer cells to a new flask or well plate
- Prepare cells for flow cytometry, assays, or cryopreservation
What are the main methods of detaching adherent cells?
The most common approaches for detaching adherent cells are enzymatic detachment (trypsin/EDTA or gentler methods), non-enzymatic detachment, and mechanical detachment (Table 1).3 The most appropriate cell detachment method depends on cell sensitivity and the downstream application. Trypsin/EDTA is commonly used for routine passaging of robust cells, while alternative methods may be preferred depending on cell sensitivity and downstream requirements (Figure 1).
| Method | Examples | Advantages | Limitations |
|---|---|---|---|
| Enzymatic (trypsin/EDTA) | Trypsin/EDTA, TrypLE | Fast, widely used, reproducible | Can damage surface proteins; requires neutralization |
| Enzymatic (mild) | Accutase-Solution | Gentler; better surface marker preservation | Higher cost; may be slower for some cell lines |
| Non-enzymatic | EDTA-only, Macrophage Detachment Solution | No protease activity; preserves epitopes | Less effective for strongly adherent cells |
| Mechanical | Cell scraper, shaking | No reagents needed; fast | Risk of physical damage; poor viability |
Table 1: Advantages and limitations of different methods for dettaching adherent cells.1,3,4
Cell detachment is part of a broader primary cell culture workflow. For additional guidance on handling primary cells, including thawing, subculturing, and maintaining cell viability, see our Best practices for primary cell culture resource.
How to choose the right cell detachment method
In practice, the most appropriate cell detachment method depends on both cell sensitivity and downstream application requirements:
- Trypsin/EDTA is commonly used for routine passaging of robust, easy-to-detach adherent cells.
- Accutase is typically preferred for sensitive primary cells or when preserving surface proteins and receptor epitopes is important.
- Non-enzymatic detachment solutions may be used when enzymatic activity could interfere with downstream assays or alter cell phenotype.
- Mechanical detachment is generally avoided unless no alternative is suitable, as it can reduce cell viability and cause damage.
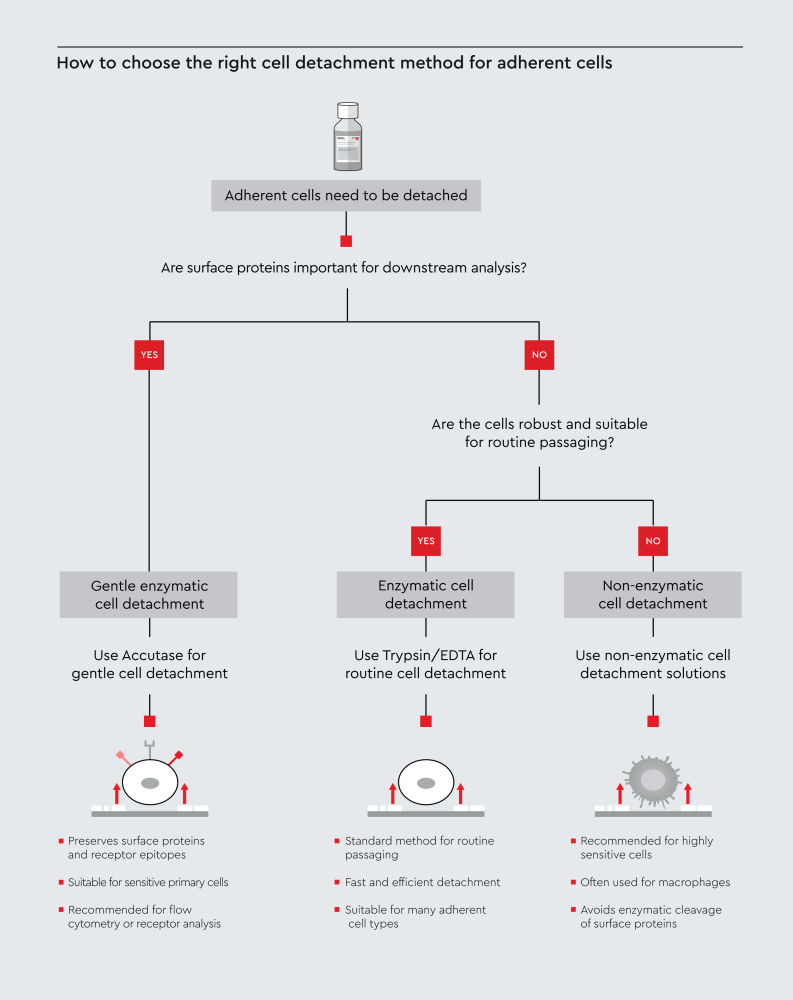

Figure 1: Decision workflow illustrating how to select an appropriate cell detachment method for adherent cells based on cell sensitivity and downstream experimental requirements, including surface protein preservation. The workflow highlights common approaches including Trypsin/EDTA, Accutase, and non-enzymatic detachment solutions.
What is trypsinization and how does trypsin detach cells?
Trypsinization is a widely used enzymatic method for harvesting adherent cells. It uses trypsin, a serine protease, to cleave the adhesion proteins that anchor cells to the culture surface.5 Trypsinization detaches cells and allows you to collect them as a suspension for passaging or downstream applications.
In cell culture, trypsin is typically used in combination with EDTA as a trypsin/EDTA solution, which enhances detachment efficiency for many adherent cell types.5 This approach provides a reliable and reproducible method for routine cell harvesting in standard cell culture workflows. Trypsinization is therefore most suitable for routine detachment workflows where speed and efficiency are required, and preservation of surface proteins is not the primary concern.
Our Trypsin/EDTA cell detachment solutions offer consistent performance across a wide range of adherent cell types.
The efficiency of trypsin/EDTA-based cell detachment depends on both trypsin activity and the presence of EDTA, which further weakens cell adhesion and facilitates cell release from the culture surface.6,7
How is trypsin activity measured?
Trypsin solutions can be specified either by percentage concentration or by enzymatic activity. One commonly used activity measure is BAEE units (BAEE-U/ml), which quantify how efficiently trypsin cleaves a defined substrate under standardized conditions.
Activity-based specifications provide a direct indication of enzyme performance and allow consistent comparison between batches. For example, an activity of 700 BAEE-U/ml corresponds to the enzymatic activity of a typical 0.04% trypsin solution used for cell detachment.
What is the role of EDTA in trypsinization?
EDTA improves cell detachment by binding calcium and magnesium ions in the culture medium.6,7 Many cell adhesion molecules depend on these ions to maintain stable interactions with the culture surface.
By removing these divalent cations, EDTA weakens cell–surface and cell–cell adhesion. This makes cells easier to detach when exposed to trypsin.8,9 For this reason, trypsin is commonly used as a trypsin/EDTA solution, where EDTA reduces adhesion while trypsin cleaves adhesion proteins. The combination allows faster and more efficient cell dissociation while helping preserve cell viability.
Troubleshooting checklist
- Cells appear rounded but don’t detach properly: It may indicate that the trypsin concentration is too low, or the incubation is too short.
- Low cell viability post-detachment: Check trypsin incubation time and neutralization step.
- Poor results in flow cytometry: Surface markers may have been cleaved; consider switching to a gentler detachment solution such as Accutase.
- Cell clumping after detachment: Pipette gently to dissociate; check that neutralization was complete.
- Prevention tips: Warm reagents to the recommended temperature before use; monitor cells under the microscope; stop trypsin activity promptly using Trypsin Neutralizing Solution.
What happens if you over-trypsinize cells?
Over-trypsinization is one of the most common mistakes in cell detachment workflows. Leaving trypsin on cells for too long or using excessive enzyme concentrations can damage cells and affect downstream experiments.5
Effects of over-trypsinization
Over-trypsinization can lead to5,10–12:
- Reduced cell viability and increased cell death
- Loss of surface proteins and receptor epitopes, compromising downstream assays such as flow cytometry
- Membrane damage leading to cell lysis
- Cell clumping due to incomplete dissociation or re-aggregation
- Changes in gene expression and cellular behavior
Troubleshooting checklist
- Cells appear rounded but don’t detach properly: It may indicate that the trypsin concentration is too low, or the incubation is too short.
- Low cell viability post-detachment: Check trypsin incubation time and neutralization step.
- Poor results in flow cytometry: Surface markers may have been cleaved; consider switching to a gentler detachment solution such as Accutase.
- Cell clumping after detachment: Pipette gently to dissociate; check that neutralization was complete.
- Prevention tips: Warm reagents to the recommended temperature before use; monitor cells under the microscope; stop trypsin activity promptly using Trypsin Neutralizing Solution.
Effects of over-trypsinization
Over-trypsinization can lead to5,10–12:
- Reduced cell viability and increased cell death
- Loss of surface proteins and receptor epitopes, compromising downstream assays such as flow cytometry
- Membrane damage leading to cell lysis
- Cell clumping due to incomplete dissociation or re-aggregation
- Changes in gene expression and cellular behavior
Troubleshooting checklist
- Cells appear rounded but don’t detach properly: It may indicate that the trypsin concentration is too low, or the incubation is too short.
- Low cell viability post-detachment: Check trypsin incubation time and neutralization step.
- Poor results in flow cytometry: Surface markers may have been cleaved; consider switching to a gentler detachment solution such as Accutase.
- Cell clumping after detachment: Pipette gently to dissociate; check that neutralization was complete.
- Prevention tips: Warm reagents to the recommended temperature before use; monitor cells under the microscope; stop trypsin activity promptly using Trypsin Neutralizing Solution.
Accutase vs trypsin: What’s the difference?
The main difference between Accutase and trypsin is that Accutase better preserves surface proteins, while trypsin provides faster and more efficient cell detachment. Accutase is a cell detachment solution containing a combination of proteolytic and collagenolytic enzymes.1 It is often used as a gentler alternative to trypsin for cell dissociation, particularly for sensitive primary cells.
Key differences between Accutase and trypsin include:
- Neutralization: Trypsin typically requires a neutralization step (e.g., adding serum-containing medium or a Trypsin Neutralizing Solution); Accutase can often be stopped simply by dilution with medium.13,14
- Cell sensitivity: Trypsin is faster and works well for robust, easy-to-detach cell lines, but Accutase may be preferred for sensitive cells (e.g., primary cells).1
- Surface marker expression: Accutase generally preserves surface markers better than trypsin, making it suitable when cells are used for flow cytometry.1,15
- Handling conditions: Both require cell detachment at room temperature and monitoring of the detachment process under a microscope.1
Our Accutase-Solution is a ready-to-use formulation developed for gentle and effective detachment of adherent cells. The balanced enzyme combination helps preserve surface proteins and epitopes, making it suitable for applications where maintaining cell surface proteins unchanged is important.
However, the most appropriate cell detachment method always depends on the cell type, culture conditions, and requirements of the downstream experiment.
How can you detach cells without damaging them?
The key to damage-free cell detachment is minimizing enzymatic exposure time and handling cells gently at every step. Below, we provide a practical step-by-step workflow for trypsin-based cell detachment.
Step-by-step cell detachment protocol using Trypsin/EDTA:
-
- Place Trypsin/EDTA (ready-to-use) solution and Trypsin Neutralizing Solution at room temperature for at least 30 minutes to adjust the temperature of the reagents.
- Remove the culture medium and rinse cells with pre-warmed PBS or balanced salt solution. This removes residual serum, which can inhibit trypsin.
- Add the appropriate volume of Trypsin/EDTA solution to fully cover the cell layer. As a guideline, use approximately 100 µl per cm² of vessel surface.
- Incubate at room temperature and check cells every 1–2 minutes under the microscope. Don’t leave unattended. Most cell lines detach within 2–5 minutes.
- Once cells round up and detach, immediately add Trypsin Neutralizing Solution or serum-containing medium (at least 3× the volume of trypsin used) to stop enzymatic activity.
- Transfer the cell suspension to a centrifuge tube, pellet the cells, and resuspend them in fresh medium.
For more details on how to detach cells using our Trypsin/EDTA solution without damaging the cells, see our instruction manual.
Our Trypsin Neutralizing Solution is formulated to reliably stop trypsin activity and protect cells after detachment. For sensitive primary cells, consider replacing trypsin with Accutase solution for gentler dissociation.
When are non-enzymatic detachment solutions recommended?
Non-enzymatic cell detachment may be the right choice when enzymatic activity itself poses a risk. This is particularly relevant when enzymatic treatment could alter cell phenotype, affect surface proteins, or interfere with downstream assays.3
Non-enzymatic detachment solutions typically rely on chelating agents (such as EDTA) to disrupt calcium-dependent adhesion without any protease activity.1,3 This approach is particularly useful for:
- Macrophages and monocyte-derived cells, which are difficult to detach without activation or damage.12,16
- Cells destined for highly sensitive receptor studies where even mild trypsin activity could alter results.1
- Cells on certain substrates where enzymatic reagents are known to interfere.17
Studies have shown that enzymatic detachment methods affect the phenotype and function of macrophages.12,16 Our non-enzymatic detachment solutions for sensitive adherent cells, including the Macrophage Detachment Solution, offer a practical enzyme-free option for these challenging workflows.
However, non-enzymatic methods are generally slower and less effective for strongly adherent cell types. The right approach always depends on the biology of your cells and the sensitivity of your downstream assays.
Need help choosing the right cell detachment method?
Selecting the optimal cell detachment method depends on your specific cell type and application. If you need support choosing the right solution for your workflow, feel free to contact our team of experts.
References
Expand
- Lai TY, Cao J, Ou-Yang P, et al. Different methods of detaching adherent cells and their effects on the cell surface expression of Fas receptor and Fas ligand. Sci Rep. 2022;12(1):5713. doi:10.1038/s41598-022-09605-y
- Mahabadi S, Labeed FH, Hughes MP. Effects of cell detachment methods on the dielectric properties of adherent and suspension cells. ELECTROPHORESIS. 2015;36(13):1493-1498. doi:10.1002/elps.201500022
- Heng BC, Cowan CM, Basu S. Comparison of enzymatic and non-enzymatic means of dissociating adherent monolayers of mesenchymal stem cells. Biol Proced Online. 2009;11(1):161. doi:10.1007/s12575-009-9001-4
- Leisi Mehrabani F, Alibeigian Y, Baghaban Eslaminejad M, Hosseini S. Mechanical harvesting of cell sheets: an efficient approach for bone and cartilage tissue engineering. Stem Cell Res Ther. 2025;16(1):310. doi:10.1186/s13287-025-04411-5
- Lordon B, Campion T, Gibot L, Gallot G. Impact of trypsin on cell cytoplasm during detachment of cells studied by terahertz sensing. Biophys J. 2024;123(16):2476-2483. doi:10.1016/j.bpj.2024.06.011
- George T, Brady MF. Ethylenediaminetetraacetic Acid (EDTA). In: StatPearls. StatPearls Publishing; 2025. Accessed February 24, 2026. http://www.ncbi.nlm.nih.gov/books/NBK565883/
- Karimi F, O’Connor AJ, Qiao GG, Heath DE. Integrin clustering matters: a review of biomaterials functionalized with multivalent integrin‐binding ligands to improve cell adhesion, migration, differentiation, angiogenesis, and biomedical device integration. Adv Healthc Mater. 2018;7(12):1701324. doi:10.1002/adhm.201701324
- Fong D, Duceppe N, Hoemann CD. Mesenchymal stem cell detachment with trace trypsin is superior to EDTA for in vitro chemotaxis and adhesion assays. Biochem Biophys Res Commun. 2017;484(3):656-661. doi:10.1016/j.bbrc.2017.01.171
- Corver WE, Cornelisse CJ, Hermans J, Fleuren GJ. Limited loss of nine tumor‐associated surface antigenic determinants after tryptic cell dissociation. Cytometry. 1995;19(3):267-272. doi:10.1002/cyto.990190311
- Huang HL, Hsing HW, Lai TC, et al. Trypsin-induced proteome alteration during cell subculture in mammalian cells. J Biomed Sci. 2010;17(1):36. doi:10.1186/1423-0127-17-36
- Reichard A, Asosingh K. Best practices for preparing a single cell suspension from solid tissues for flow cytometry. Cytometry A. 2019;95(2):219-226. doi:10.1002/cyto.a.23690
- Chen S, So EC, Strome SE, Zhang X. Impact of detachment methods on M2 macrophage phenotype and function. J Immunol Methods. 2015;426:56-61. doi:10.1016/j.jim.2015.08.001
- Brown MA, Wallace CS, Anamelechi CC, Clermont E, Reichert WM, Truskey GA. The use of mild trypsinization conditions in the detachment of endothelial cells to promote subsequent endothelialization on synthetic surfaces. Biomaterials. 2007;28(27):3928-3935. doi:10.1016/j.biomaterials.2007.05.009
- Matinfar A, Dezfulian M, Haghighipour N, Kurdtabar M, Pourbabaei AA. Replacement of trypsin by proteases for medical applications. Iran J Pharm Res. 2022;21(1). doi:10.5812/ijpr-126328
- Mutin M, George F, Lesaule G, Sampol J. Reevaluation of trypsin-EDTA for endothelial cell detachment before flow cytometry analysis. Endothelium. 1996;4(4):289-295. doi:10.3109/10623329609024704
- Nielsen MC, Andersen MN, Møller HJ. Monocyte isolation techniques significantly impact the phenotype of both isolated monocytes and derived macrophages in vitro. Immunology. 2020;159(1):63-74. doi:10.1111/imm.13125
- Kovács KD, Szittner Z, Magyaródi B, et al. Optical sensor reveals the hidden influence of cell dissociation on adhesion measurements. Sci Rep. 2024;14(1):11719. doi:10.1038/s41598-024-61485-6
Related resources
