3D Cell Culture
Find suitable products, scientific resources or answers to your questions within our technical library FAQ.
Checkout using your account
This form is protected by reCAPTCHA - the Google Privacy Policy and Terms of Service apply.
Checkout as a new customer
Creating an account has many benefits:
Mesenchymal stem cells (MSCs) are multipotent adult stem cells found in connective tissue. They are often also referred to as “mesenchymal stromal cells” by the scientific community.1 MSCs are adherent, fibroblast-like cells with regenerative properties.2,3 They also release cytokines and extracellular vesicles that communicate with surrounding cells to maintain tissue homeostasis.
Human MSCs show promise in regenerative medicine and stem cell therapy for treating autoimmune diseases, inflammatory conditions, and tissue damage.2,3 The therapeutic value of MSCs stems from their ability to regulate immune responses and support tissue repair through paracrine signaling.
Explore our cell culture basics page on MSCs to learn more about MSC biology.
Mesenchymal stem cells can be isolated from several tissue sources in the body. Common sources include the bone marrow, adipose tissue, and umbilical cord tissues like Wharton’s jelly.4,5 Other sources include peripheral blood (although MSCs are found at a lower frequency), dental pulp, and placental tissues.4,6
Isolation of MSCs is based on enrichment of stromal stem cell populations through various techniques:
Large-scale isolation from human bone marrow requires significant time and resources, and its success depends on bone marrow sample quality and donor age.10
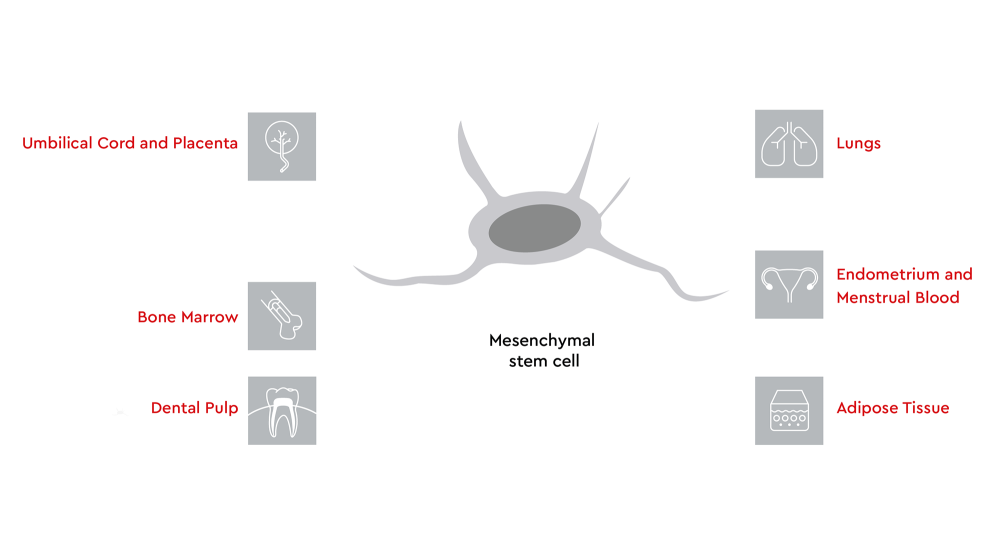

Figure 1: Sources of mesenchymal stem cells in the human body. Tissue sources of MSCs include umbilical cord and placenta, bone marrow, dental pulp, lungs, endometrium and menstrual blood, and adipose tissue.
Mesenchymal stem cells require a controlled culture environment, including the selection of an optimized MSC cell culture medium, to ensure that their biological properties are reproducible.11 Serum can support cell growth, but it adds variability. Fetal bovine serum is complex and changes from lot to lot, which can influence the proliferation, morphology, and functional characteristics of cells.11,12 Defined or reduced-serum media, such as xeno-free MSC culture systems, reduce variability and increase reproducibility.
Low-serum or serum-free media have the benefit of providing more standardized culture conditions over a long time span. Examples of low-serum or serum-free options include Mesenchymal Stem Cell Growth Medium XF, Mesenchymal Stem Cell Growth Medium 2, and the Excipient GMP-grade PromoExQ MSC Growth Medium XF (prf).
The use of supplements, the choice of extracellular matrix, and oxygen tension in cell culture can also influence mesenchymal stem cell characteristics. For example, extracellular matrix and oxygen tension can affect adhesion properties, proliferation kinetics, and differentiation potential of MSCs.13,14
Passaging practices can also influence MSC quality and expansion. When using our Mesenchymal Stem Cell Growth Medium 2, seeding at 5,000 cells/cm² typically allows cultures to reach 80%–90% confluency within 4–6 days. High passage numbers can lead to reduced proliferation, altered morphology, and genetic changes that could compromise the therapeutic potential of MSCs.15,16
Producing high-quality mesenchymal stem cells in sufficient quantities for therapeutic applications is challenging. Mesenchymal stem cells make up only 0.001%–0.01% of mononuclear cells in bone marrow from adult donors.17 Regenerative medicine applications require infusions containing several million cells per kilogram of body weight.18 MSCs are sensitive to their environment, and suboptimal conditions can lead to phenotypic drift, reduced proliferation, and morphological changes.17
The use of consistent, optimized protocols is key for successful scale-up of MSC cultures for clinical applications. Standardized procedures help maintain cell quality across production batches.17,18 Process controls monitor parameters like cell density, medium composition, and environmental conditions. Quality assurance testing at multiple stages confirms that expanded populations retain their therapeutic properties and meet safety standards.17,18
Long-term storage through cryopreservation allows MSC banking for future use. Nontoxic, xeno-free cryopreservation methods protect cell viability and function during freezing and thawing.19,20 For researchers requiring reliable cryopreservation solutions, Freezing Medium Cryo-SFM Plus (for GMP environment: PromoExQ CellNAP) offers a serum-free option that maintains MSC viability and phenotype during storage.
We at PromoCell offer several mesenchymal stem cell products that support high-quality MSCs and reproducible performance for both research and clinical applications. Our portfolio includes primary cells, culture media, and differentiation media.
| Product | Size | Cat. No. |
|---|---|---|
| Mesenchymal Stem Cell Growth Medium 2 (Ready-to-use) | 500 ml | C-28009 |
| Mesenchymal Stem Cell Growth Medium 2 (Ready-to-use), phenol red-free | 500 ml | C-28017 |
| Mesenchymal Stem Cell Growth Medium XF * (Ready-to-use) | 500 ml | C-28019 |
| Mesenchymal Stem Cell Growth Medium XF * (Ready-to-use), phenol red-free | 500 ml | C-28018 |
| PromoExQ MSC Growth Medium XF * (Manufactured in compliance with the EXCiPACT™ GMP certification standard) | 500 ml | EQ-C-28019 |
Table 1. MSC isolation and expansion media
* Fibronectin- or vitronectin-coated plates are necessary in conjunction with the xeno-free (XF) media
| Product | Size | Cat. No. |
|---|---|---|
| MSC Adipogenic Differentiation Medium 2 (Ready-to-use) | 100 ml | C-28016 |
| Mesenchymal Stem Cell Chondrogenic Differentiation Medium (Ready-to-use) with or without inducers | 100 ml each | C-28012/C-28014 |
| Mesenchymal Stem Cell Osteogenic Differentiation Medium (Ready-to-use) | 100 ml | C-28013 |
| Mesenchymal Stem Cell Neurogenic Differentiation Medium (Ready-to-use) | 100 ml | C-28015 |
Table 2. MSC differentiation media
For project-specific guidance on culture optimization or differentiation protocols, contact our scientific support team.
References
Expand
Have a look at other Research Areas
PromoCell uses HubSpot to provide LiveChat support. This feature is currently blocked due to your cookie preferences.
Accept cookies to enable LiveChat