EXCiPACT™ GMP certified produced cell culture media
A crucial step when developing new drugs and cell therapies and during development of new cosmetics or cell-based food production, is the translation of a technology from the research laboratory to clinical use in the real world, and subsequently commercial manufacturing. To ensure this step goes well, understanding and evaluating potential raw material risks of media and reagents is key when working with products in a regulated environment and regulatory agencies such as U.S. Food and Drug Administration. Therefore, to mitigate such risks, PromoCell offers Excipient GMP-grade* cell culture media that are manufactured in an EXCiPACT certified environment.
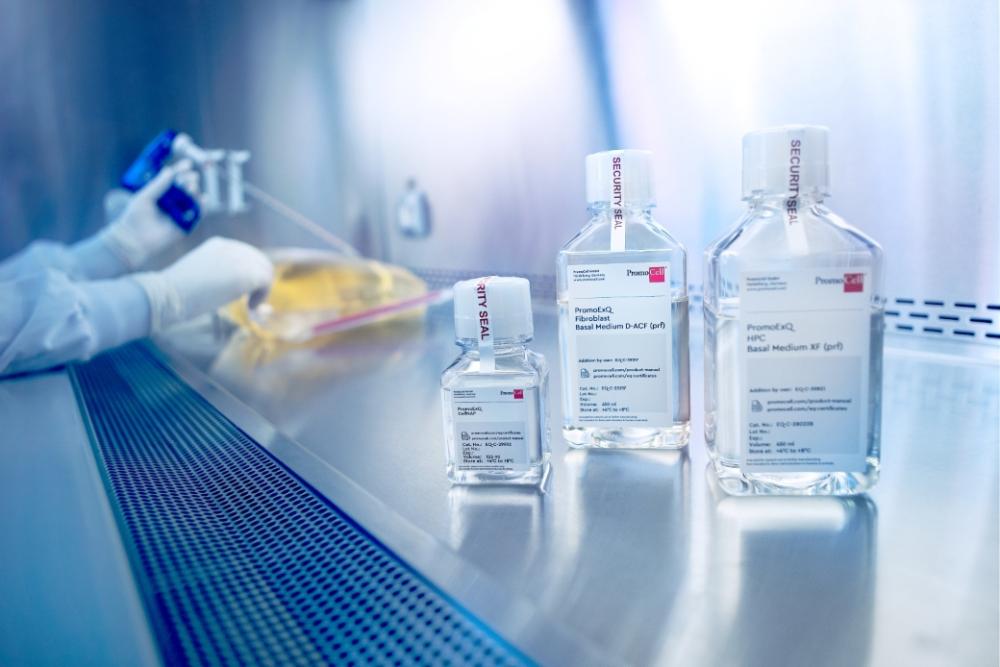

Why Excipient GMP-grade* media?
To ensure the transition from bench to commercial manufacture goes smoothly when developing cell-based therapies, the earliest stages of preclinical research should include cell culture media and reagents that are manufactured in compliance with International Pharmaceutical Excipients Council (IPEC) international Good Manufacturing Practice (GMP) guidelines.
GMP-grade* cell culture media are vital for restraining safety and quality, complying with strict standards, supporting cell viability, functionality, and consistency in manufacturing processes.
Our entire product development pipeline is designed to enable compliance with these regulatory guidelines. We are proud to support you from the first step in preclinical development to development of the final therapy.
Our Commitment to Quality
Our Quality Management System is certified to the EXCiPACT™ GMP standard. The EXCiPACT™ certification framework for pharmaceutical excipients and Pharmaceutical Auxiliary Materials (PAMs) builds on our existing ISO 9001:2015 quality management system. It provides a GMP standard for pharmaceutical excipients or PAMs that is accepted worldwide.
What is Excipient GMP-grade*?
The EXCiPACT™ GMP certification covers the entire production process for our cell culture media and reagents (EQ-C-*/PE-C-*), ensuring a standardized environment, controlled equipment, and qualified raw materials. With the extended scope to include excipients and pharmaceutical auxiliary materials (PAMs), we have established a robust quality framework for materials indirectly used in pharmaceutical manufacturing, such as processing aids and buffers. This standard integrates relevant regulatory and quality requirements and thereby ensures consistent quality, safety, and regulatory compliance of excipients and PAMs. Therefore, this certification also ensures that our media undergo stringent quality control before product release. In addition to the EXCiPACT™ GMP certification, we also provide relevant documentation to support research in a regulated environment.
| Material category | What it is | Included in the final drug product? | Intentional patient contact? | Compliance framework | Typical examples |
|---|---|---|---|---|---|
| Pharmaceutical Auxiliary Materials (PAMs) | Processing materials used to manufacture APIs and medicinal products. | No (residual levels might be possible) | No | EXCiPACT PAMs Standard | Solvents, processing aids, cell culture media, pharmaceutical gases, etc. |
| Medical Devices | Instruments, implants, or in vitro tools/reagents used to diagnose, monitor, prevent, or treat health conditions. | Administration devices (e.g., syringes) are not included; cell therapies where cells are administered are included. | Yes | 21 CFR 820; ISO 13485 | Syringes, catheters, pacemakers, ex vivo grown cells reintroduced into patients, etc. |
| Pharmaceutical Excipients | Non active ingredients added to medicines to aid, improve stability and bioavailability, enhance patient acceptability, support delivery, and preserve product integrity during storage. | Yes | Yes | IPEC PQG; EXCiPACT Standard | Fillers, carriers that aid API delivery to the patient, etc. |
| Active Pharmaceutical Ingredients (APIs) | The pharmacologically active substance responsible for the medicine’s therapeutic activity. | Yes | Yes | 21 CFR 210/211; EudraLex; ICH / OECD Guidelines | Antibody-drug conjugates, monoclonal antibodies, etc. |
| Material category | What it is | Included in the final drug product? | Intentional patient contact? | Compliance framework | Typical examples |
|---|---|---|---|---|---|
| Pharmaceutical Auxiliary Materials (PAMs) | Processing materials used to manufacture APIs and medicinal products. | No (residual levels might be possible) | No | EXCiPACT PAMs Standard | Solvents, processing aids, cell culture media, pharmaceutical gases, etc. |
| Medical Devices | Instruments, implants, or in vitro tools/reagents used to diagnose, monitor, prevent, or treat health conditions. | Administration devices (e.g., syringes) are not included; cell therapies where cells are administered are included. | Yes | 21 CFR 820; ISO 13485 | Syringes, catheters, pacemakers, ex vivo grown cells reintroduced into patients, etc. |
| Pharmaceutical Excipients | Non active ingredients added to medicines to aid, improve stability and bioavailability, enhance patient acceptability, support delivery, and preserve product integrity during storage. | Yes | Yes | IPEC PQG; EXCiPACT Standard | Fillers, carriers that aid API delivery to the patient, etc. |
| Active Pharmaceutical Ingredients (APIs) | The pharmacologically active substance responsible for the medicine’s therapeutic activity. | Yes | Yes | 21 CFR 210/211; EudraLex; ICH / OECD Guidelines | Antibody-drug conjugates, monoclonal antibodies, etc. |
Research use only
vs regulated products
The specifications and documents available for research use only (C-*)
and regulated products (EQ-C-* | PE-C-*).
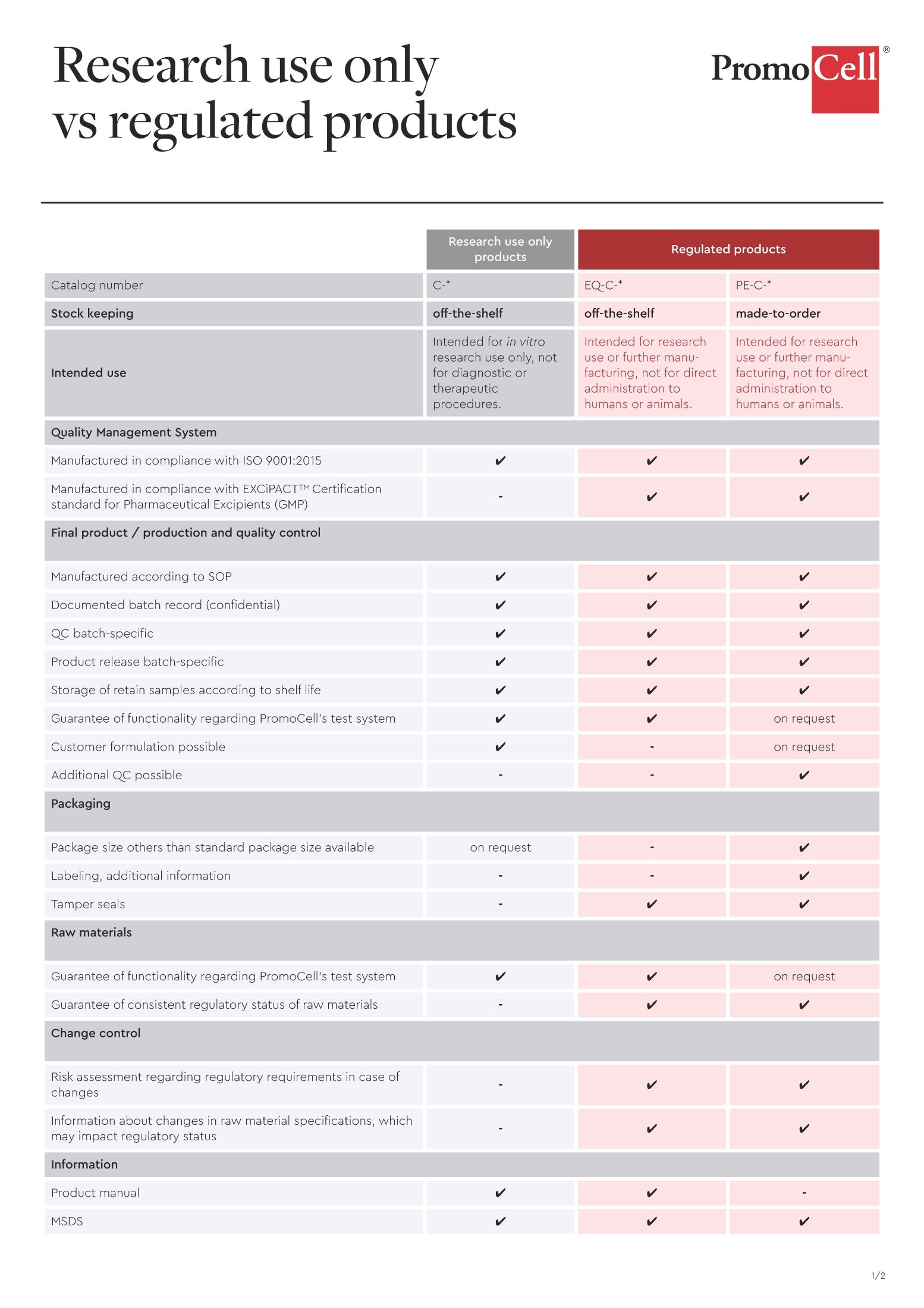

*”GMP-grade” is a branding term used by PromoCell to denote reagents that are manufactured at the PromoCell manufacturing facility in Heidelberg, Germany, under strictly controlled processes to meet stringent product specifications and customer requirements. Reagents manufactured at PromoCell are produced according to EXCiPACT™ GMP standards, a quality management system that builds on our ISO 9001:2015 certification. Risk assessment procedures are carried out at the customer site.
This material was initially created by RegMedNet and the original version can be found here.
FAQ
