Stem cell cryopreservation is the controlled freezing of stem cells to preserve their viability and function for future research or therapeutic use. It is one of the most important steps in cell-based research or therapy workflows. This blog article explores the main challenges of stem cell cryopreservation and practical steps to improve cell viability.
At a glance: Quick takeaways
Why is protocol optimization essential for stem cell cryopreservation?
Stem cells are highly sensitive to freeze-thaw stress, and using optimized protocols can improve post-thaw viability and function.What ensures consistent stem cell cryopreservation?
Using a serum-free, defined, and CPA-optimized cryopreservation medium is one of the most important factors for consistent stem cell cryopreservation.What affects stem cell recovery after thawing?
Post-thaw stem cell recovery depends on controlled thawing speed, gentle dilution, and optimized plating conditions.What is Excipient GMP-grade* cryopreservation media?
or GMP-regulated workflows, using Excipient GMP-grade* cryopreservation media ensures consistency, safety, and regulatory alignment.How to cryopreserve stem cells
The stem cell cryopreservation protocol follows five key steps.
- Harvest cells during the logarithmic growth phase, ensuring low culture confluence (under 80%) and minimal differentiation.
- Resuspend cells in a freezing medium containing a cryoprotectant agent (CPA) such as 10% DMSO to prevent intracellular ice crystal damage.
- Use a controlled-rate freezer or isopropanol container to achieve a cooling rate of approximately 1°C/minute.
- Transfer vials to a −80°C freezer overnight.
- Move the vials to vapor-phase liquid nitrogen for long-term storage.
Common challenges in the stem cell cryopreservation process
Stem cells are sensitive, making stem cell cryopreservation challenging.1 Several common factors can affect the viability and behavior of stem cells during freeze-thaw cycles:
- Inconsistent post-thaw viability: Cell recovery can differ from vial to vial or from batch to batch.1–3
- Functional drift: Cells may show altered morphology, reduced marker expression, or impaired differentiation after thawing.1–3
- Serum-induced variability: Lot-to-lot differences in serum-containing media can introduce unpredictability in the behavior of stem cells during freeze-thaw cycles.1–3
- Cell bank inconsistency: Master and working banks behaving differently creates delays in process development and comparability studies.1–3
Because many factors can affect the freeze-thaw viability and behavior of stem cells, small improvements in your cryopreservation protocol can make a big difference in the results you get.1–3
What causes cell damage during cryopreservation?
Cell damage during cryopreservation results from changes inside the cells that occur as the temperature drops.4 These changes include:
- Ice crystal formation: Intracellular ice crystals damage membranes and organelles, which can cause cell death during rapid freezing.4
- Osmotic stress: As extracellular water freezes, solute concentrations rise quickly. Water moves out of cells, causing shrinkage and membrane damage.4
- Oxidative damage: Freeze-thaw cycles generate reactive oxygen species (ROS), which damage proteins, lipids, and DNA.4
- Cryoprotectant (CPA) toxicity: Agents like DMSO can reduce cell damage during cryopreservation, but they become toxic at higher concentrations and temperatures.4,5
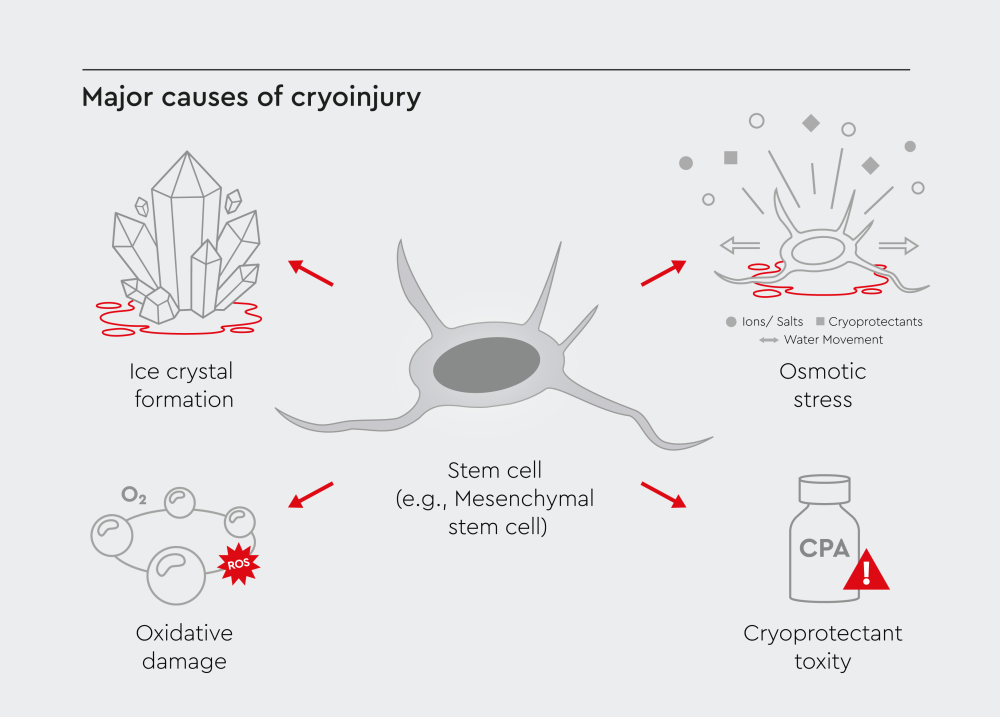

Figure 1: Key mechanisms contributing to cryoinjury, including ice crystal formation, osmotic stress, oxidative damage, and cryoprotectant toxicity
Sensitive cells such as iPSCs and adult stem cells are particularly prone to cell damage during cryopreservation.6
How can stem cell cryoinjury be minimized?
If you optimize each step of cryopreservation, you can reduce cryoinjury and achieve consistent cell viability and functional stability during long-term stem cell storage.7,8
What factors affect stem cell viability during cryopreservation?
Multiple variables interact to determine how well cells survive the freeze-thaw cycle.7,8 The table below summarizes key factors and freezing tips, including the use of serum-free formulations, to improve stem cell viability during cryopreservation.
| Factor | Why it matters | Freezing tips |
|---|---|---|
| Stem cell type and state | Different sensitivities to stress and freezing | Match method to cell type (single cells vs. clumps). Handle gently. |
| Passage number / culture history | Stressed or high-passage cells recover poorly | Freeze healthy, log-phase cultures. |
| Cooling rate | Too fast = ice crystals; too slow = dehydration | Use controlled-rate freezing (~1°C/min) for consistent results |
| Cell density | Influences CPA protection and recovery | Use a consistent, appropriate density range for your cell type. |
| Serum vs. defined media | Serum adds a lot-to-lot variability | Prefer defined, serum-free formulations for reproducibility. |
| Oxidative stress | Freeze-thaw cycles generate ROS, which damage proteins, lipids, and DNA | Use cryopreservation medium with antioxidant components to counteract oxidative stress. |
| Handling time and temperature | CPA toxicity increases with time and temperature | Work efficiently; pre-chill reagents and equipment where appropriate. |
| Storage conditions | Temperature fluctuations reduce long-term viability | Store in stable vapor-phase liquid nitrogen. |
Table 1: Key factors influencing stem cell cryopreservation
Standardizing stem cell cryopreservation is essential for reproducible cell banks and comparable downstream results for cell therapy workflows.
How can you further improve post-thaw recovery of stem cells?
Post-thaw recovery improves when every step after removing the vial from storage is as controlled and optimized:
- Thaw rapidly. Move vials quickly from liquid nitrogen to a 37°C water bath. Fast thawing limits re-crystallization of residual ice, one of the main sources of post-thaw cryoinjury.8,9
- Dilute gently. Add warm medium dropwise to reduce osmotic shock as DMSO dilutes out. A sudden osmotic shift stresses cell membranes.8,9
- Optimize plating density. Plate at a higher density than standard culture. Post-thaw cells benefit from paracrine support during initial recovery.8,9
- Time the first media change carefully. Allow cells to settle and begin recovering before the first media exchange. The optimal timing of the first media change varies by cell type.8,9
- Record recovery metrics. Measure viability (e.g., trypan blue or automated counting), recovery yield, and time to re-confluence consistently.8,9
Why is stem cell cryopreservation important for cell therapy?
Stem cell cryopreservation is essential for scaling cell-based therapies. It enables the standardized storage, transport, and on-demand availability of consistent cell batches across research, manufacturing, and cell therapy workflows.
Without stem cell cryopreservation, translating cell-based therapies (e.g., allogeneic cell therapy workflows) from the lab to the clinic becomes logistically and scientifically much harder.10 Here’s why:
- Cell banking: Establishing a well-characterized bank of cryopreserved cells helps accelerate reproducible production of cells for therapeutic purposes without the need for continuous culture.
- Comparability across lots and sites: When each batch starts from the same banked material, cross-site and cross-lot comparisons become easier.
- Off-the-shelf flexibility: Cryopreserved cells can be thawed as needed, reducing lead times for assays, process development runs, and clinical manufacturing steps.
- Reduced passage-related drift: Cryopreserving cells at a specific passage number can minimize phenotypic and functional changes that accumulate in cells with serial passaging.
- Defined QC checkpoints: Post-thaw recovery metrics (viability, marker expression, potency assays) serve as quality control measures before cells move to the next manufacturing step.
Designing a robust cryopreservation strategy requires not just a reliable protocol, but also the right medium. The composition of the freezing solution influences how well cells survive the freeze-thaw cycle.
How does cryopreservation medium affect stem cell viability?
Cryopreservation medium affects stem cell viability primarily by balancing the prevention of ice crystal formation against the chemical toxicity of protective additives. A high-quality cryopreservation medium for stem cells must therefore protect cells against multiple mechanisms of cryoinjury simultaneously, providing effective protection even under suboptimal conditions.
Key components you should look for in a cryopreservation medium include:
- DMSO: The most widely used cryoprotectant, DMSO, penetrates cells and limits intracellular ice formation. Typically used at 5%–10% in stem cell protocols.4
- Methylcellulose and complementary CPAs: These stabilize the extracellular environment during controlled cooling, reducing osmotic shock and membrane stress.4
- Antioxidants: Freeze-thaw cycles generate reactive oxygen species. Antioxidant components in the medium can reduce oxidative damage.11
- Defined, serum-free formulation: Serum introduces animal-derived material and lot-to-lot variability, reducing reproducibility and hindering GMP-aligned manufacturing.12
Additionally, in GMP-regulated environments, cryopreservation requires the use of GMP-compliant manufactured cryopreservation media to ensure consistency, safety, and regulatory alignment.
When should you use research-grade vs. Excipient GMP-grade* cryopreservation media?
During early research and discovery, research use only (RUO) cryopreservation media are generally appropriate for protocol optimization, cell line development, and proof of concept studies where flexibility and rapid iteration are key. As a project progresses toward translational development, cell banking, or clinical manufacturing, transitioning to an Excipient GMP-grade* cryopreservation medium becomes essential. GMP-grade manufactured media are designed to meet GMP expectations for quality, documentation, and risk mitigation, including raw material traceability and consistent batch-to-batch performance. Using GMP-compliant manufactured cryopreservation media helps you minimize regulatory and comparability risks when moving from research to therapeutic application.
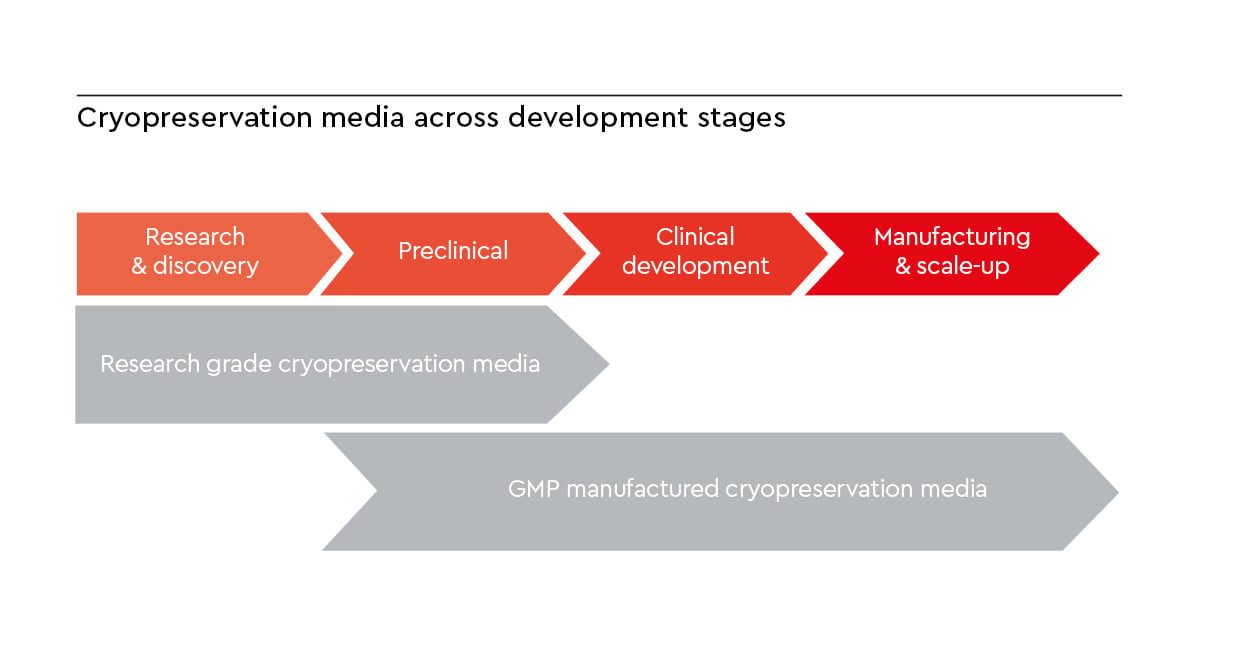

Figure 2: Developmental pathway highlighting the transition from research-grade to GMP-manufactured cryopreservation media.
How we at PromoCell support your cryopreservation, from research to GMP
Cryo-SFM Plus is an animal component-free, protein-free, defined freezing medium featuring a patented antioxidant formula that addresses freeze-thaw oxidative damage. It’s designed for sensitive stem cell workflows, including iPSC and MSC applications. Cryo-SFM Plus supports reproducible post-thaw recovery in both research and translational settings. In head-to-head comparisons, Cryo-SFM Plus outperforms leading competitor freezing media for primary human MSCs.
PromoExQ CellNAP cell freezing medium is manufactured in compliance with the EXCiPACT™ GMP certification scheme and supports cryopreservation workflows aligned with GMP requirements. It is supplied with the documentation and quality standards required for further manufacturing.
*“GMP-grade” is a branding term used by PromoCell to denote reagents that are manufactured at the PromoCell manufacturing facility in Heidelberg, Germany, under strictly controlled processes to meet stringent product specifications and customer requirements. Reagents manufactured at PromoCell are produced in accordance with EXCiPACT™ GMP standards, a quality management system that builds on our ISO 9001:2015 certification. Risk assessment procedures are carried out at the customer site.
FAQs
References
Expand
- Wang Y, Yang K, Yuan S, Huo F, Yang C, Tian W. Research advances in cryopreserved preparations of mesenchymal stem cells: technical innovations, application challenges, and quality control. Front Bioeng Biotechnol. 2026;14:1717539. doi:10.3389/fbioe.2026.1717539
- Heng BC, Ye CP, Liu H, et al. Loss of viability during freeze–thaw of intact and adherent human embryonic stem cells with conventional slow-cooling protocols is predominantly due to apoptosis rather than cellular necrosis. J Biomed Sci. 2006;13(3):433-445. doi:10.1007/s11373-005-9051-9
- Cottle C, Porter AP, Lipat A, et al. Impact of cryopreservation and freeze-thawing on therapeutic properties of mesenchymal stromal/stem cells and other common cellular therapeutics. Curr Stem Cell Rep. 2022;8(2):72-92. doi:10.1007/s40778-022-00212-1
- Whaley D, Damyar K, Witek RP, Mendoza A, Alexander M, Lakey JR. Cryopreservation: An overview of principles and cell-specific considerations. Cell Transplant. 2021;30:0963689721999617. doi:10.1177/0963689721999617
- Best BP. Cryoprotectant toxicity: Facts, issues, and questions. Rejuvenation Res. 2015;18(5):422-436. doi:10.1089/rej.2014.1656
- Hunt CJ. Cryopreservation of human stem cells for clinical application: A review. Transfus Med Hemotherapy. 2011;38(2):107-123. doi:10.1159/000326623
- Mohamed HM, Sundar P, Ridwan NAA, et al. Optimisation of cryopreservation conditions, including storage duration and revival methods, for the viability of human primary cells. BMC Mol Cell Biol. 2024;25(1):20. doi:10.1186/s12860-024-00516-6
- Uhrig M, Ezquer F, Ezquer M. Improving cell recovery: Freezing and thawing optimization of induced pluripotent stem cells. Cells. 2022;11(5):799. doi:10.3390/cells11050799
- Xie J, Ekpo MD, Xiao J, et al. Principles and protocols for post-cryopreservation quality evaluation of stem cells in novel biomedicine. Front Pharmacol. 2022;13:907943. doi:10.3389/fphar.2022.907943
- Meneghel J, Kilbride P, Morris GJ. Cryopreservation as a key element in the successful delivery of cell-based therapies—A review. Front Med. 2020;7:592242. doi:10.3389/fmed.2020.592242
- Len JS, Koh WSD, Tan SX. The roles of reactive oxygen species and antioxidants in cryopreservation. Biosci Rep. 2019;39(8):BSR20191601. doi:10.1042/BSR20191601
- Weber T, Malakpour-Permlid A, Chary A, et al. Fetal bovine serum: how to leave it behind in the pursuit of more reliable science. Front Toxicol. 2025;7:1612903. doi:10.3389/ftox.2025.1612903
- Bennett B, Hanotaux J, Pasala AR, et al. Impact of lower concentrations of dimethyl sulfoxide on cryopreservation of autologous hematopoietic stem cells: a systematic review and meta-analysis of controlled clinical studies. Cytotherapy. 2024;26(5):482-489. doi:10.1016/j.jcyt.2024.02.006
Related resources
