CD34⁺ hematopoietic progenitor cells (HPCs) are the proliferative intermediates that drive day-to-day renewal of the blood and immune cells. Arising from hematopoietic stem cells, HPCs have limited self-renewal but a robust capacity to expand and differentiate into lineage-restricted myeloid and lymphoid progeny. Read on to learn about the role of CD34⁺ HPCs and other CD34+ populations and how to expand them.
What is CD34?
CD34 is a transmembrane sialomucin protein that regulates cell adhesion and migration.1,2 CD34 is commonly used as a marker to identify hematopoietic stem and progenitor cells (HSPCs), which are the reservoir of mature blood cell types throughout the lifetime.1
CD34 is not exclusively hematopoietic; it is also expressed on several progenitor populations throughout the body.1 Although CD34+ cells are generally not pluripotent, they are multipotent to generate multiple cell types within a specific lineage.3 For example, CD34+ hematopoietic stem cells (HSCs) can produce all blood and immune cell types but cannot become neurons or muscle cells.4
The uses and biological functions of CD34 include:
- Transplantation: CD34 is used as a marker to identify and enrich human hematopoietic stem cells for bone marrow transplantation.2
- Adhesion molecule: CD34 binds to vascular selectins, regulating stem cell homing to bone marrow microvasculature.2,5
- Progenitor marker: In addition to HSPCs, CD34 is also expressed in endothelial progenitors and other tissue-specific stem cell populations.1
What are hematopoietic stem and progenitor cells?
Hematopoietic stem and progenitor cells (HSPCs) are a rare population of cells in the bone marrow. They continuously renew themselves and generate all mature blood cells (red cells, white cells, platelets) to maintain the blood system throughout life.6
Blood cells have a relatively short lifespan. For example, red blood cells last about 120 days, and certain white blood cells survive only hours.7 HSPCs are responsible for maintaining lifelong production of mature blood cells through tightly regulated self-renewal and differentiation programs.8
Hematopoietic stem vs. progenitor cells
The terms CD34+ HSCs and progenitor cells are often used interchangeably to describe the cells that produce mature blood cells. Collectively, these are referred to as hematopoietic stem and progenitor cells (HSPCs).6 However, there are differences between these subsets regarding their origin (ontogeny), self-renewal potential, differentiation potency, and proliferative state.9
Although CD34+ HSCs typically remain quiescent at the apex of the hematopoietic hierarchy to preserve lifelong self-renewal, CD34+ HPCs are actively cycling populations committed to differentiation.10–12 Furthermore, the broader category of CD34+ progenitor cells may also include non-hematopoietic lineages, such as endothelial progenitor cells (EPCs), which are vital for vascular repair and vasculogenesis.13
Explore our AppNote on the expansion of primitive hematopoietic progenitor cells.
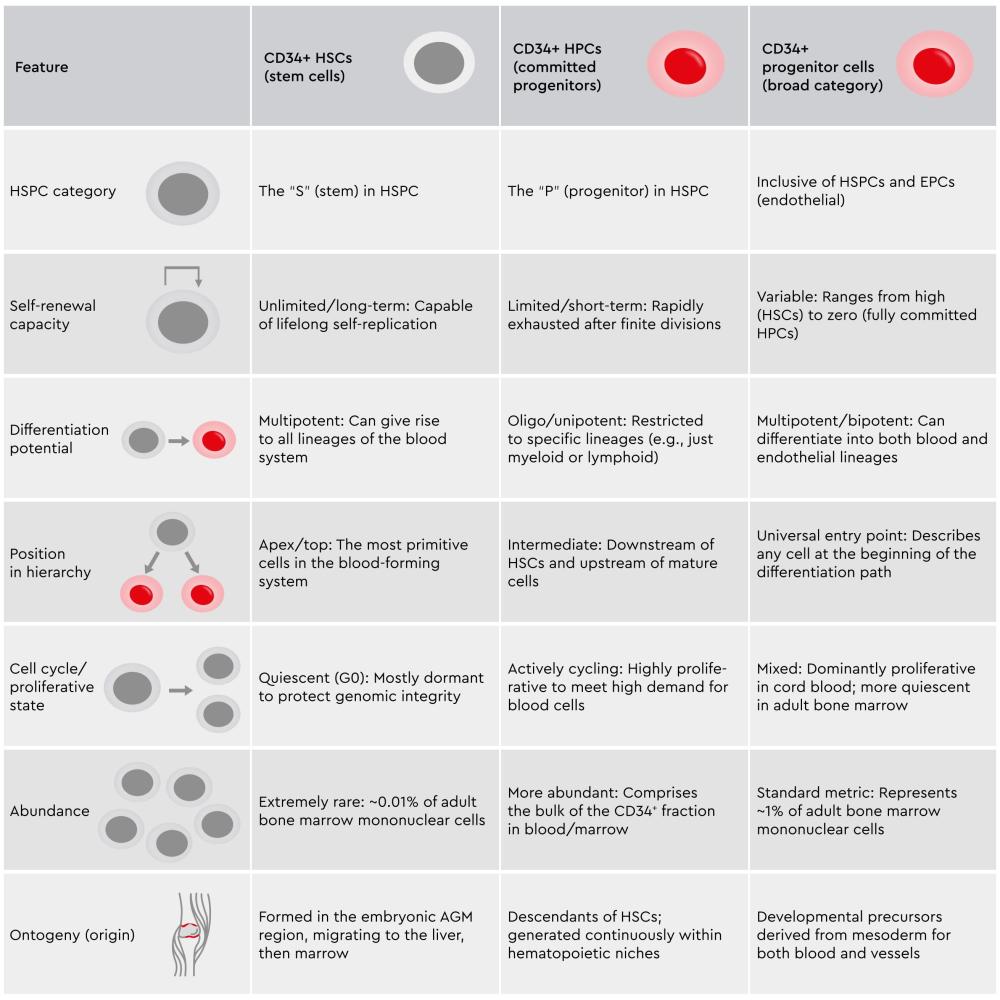

Table 1: Comparison of CD34+ cell populations in the hematopoietic hierarchy. CD34+ HSCs are the most primitive, self-renewing cells at the apex of blood formation. CD34+ HPCs are committed, actively proliferating intermediates with limited self-renewal.10,11,14–17
This hierarchical organization of hematopoietic stem and progenitor cells ensures efficient blood production, depending on the requirements. HSCs maintain the stem cell reservoir required for regenerating the entire hematopoietic system, whereas HPCs generate mature lineage-specific blood cells.18
From CD34⁺ HSPCs to immune cells
CD34+ hematopoietic stem and progenitor cells can differentiate into all types of blood and immune cells, forming the foundation of both the hematopoietic system and immune system. The differentiation of CD34+ HSPCs begins with cells committing to either a myeloid or a lymphoid lineage.18
Myeloid differentiation begins with HSC/HPC generating common myeloid progenitors (CMPs), which differentiate into granulocyte-macrophage progenitors (GMPs). Mature cells derived from GMPs include monocytes, macrophages, neutrophils, eosinophils, basophils, dendritic cells, and red blood cells. G-CSF, M-CSF, and GM-CSF are among the cytokines that promote myeloid differentiation.19,20
During lymphoid differentiation, common lymphoid progenitors (CLPs) arise from HSC/HPC. CLPs generate T lymphocytes and B lymphocytes, which form the backbone of adaptive immunity, as well as natural killer (NK) cells. IL-7 and IL-2 promote lymphoid development.19,21
The lymphoid and myeloid differentiation pathways are dynamic. During infection, cytokine signals can skew production toward immune cells. After blood loss, erythropoietin promotes the development of red blood cells.19
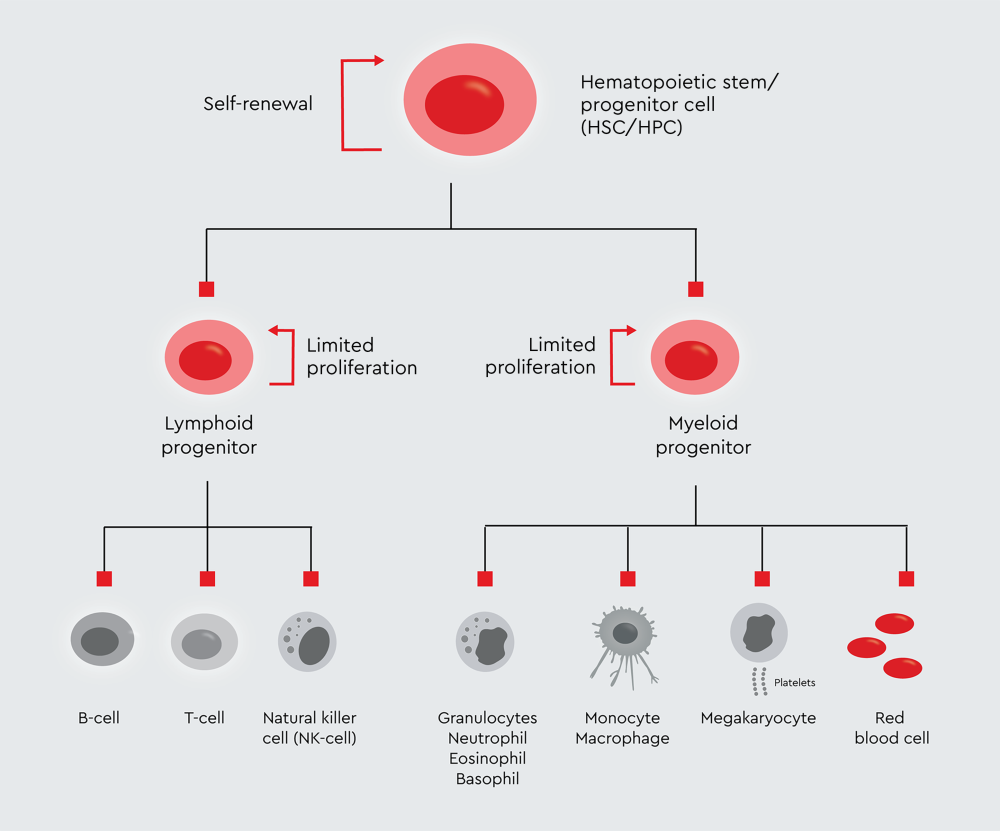

Figure 1: Overview of hematopoietic progenitor cell lineages. Schematic overview showing CD34+ hematopoietic stem and progenitor cells, branching into common myeloid progenitors (CMP) and common lymphoid progenitors (CLP). CLPs generate T cells, B cells, and NK cells. CMPs can differentiate into granulocytes, monocytes/macrophages, dendritic cells, megakaryocytes, and erythrocytes.
Applications of CD34+ cells
In research, HPCs serve as models to study how blood cells develop and differentiate into various cell types. Researchers are using CD34+ HSPCs to study hematopoiesis, stem cell aging, and stem cell dysfunction.22 Recently, researchers used CD34+ progenitors to produce therapeutic CAR-NK cells to target solid tumors and leukemias.23 Additional applications of CD34+ cells in research include screening drugs affecting blood cell development and developing organ-on-a-chip systems incorporating immune components.24
Researchers use CD34+ HSPCs as target cells for gene therapy because modifications persist throughout the patient’s lifetime.25,26 Moreover, the multipotency of CD34+ HSCs and their ability to engraft make these cells valuable for transplantation and development of gene and cell therapies.27 This makes HSPC and HSC gene therapy a one-time, potentially curative approach for inherited blood disorders, such as sickle cell disease.
Explore key media requirements to transition to cell therapy applications
Serum-free expansion of CD34+ cells
Maintaining the multipotency of CD34+ cells during cell culture is a critical challenge for translating therapies from the lab to the clinic, as cytokines and growth factors in the serum can promote premature CD34+ cell differentiation.28 Expanding CD34+ hematopoietic progenitor cells ex vivo without differentiation requires the use of serum-free, chemically defined culture systems.
Serum-free, xeno-free formulations provide:
- Consistent, defined chemical composition for reproducible expansion.
- Reduced differentiation pressure, maintaining CD34 expression and multipotency.
- Regulatory-friendly manufacturing compatible with GMP requirements.
- Enhanced ability to study specific cytokine effects.
Get insights into our cell culture media formulations
Our solutions for CD34+ cell research and manufacturing
We offer high-quality optimized cell culture media that support the complete workflow of CD34+ cell expansion, from research to clinical-grade cell therapy.
- Hematopoietic Progenitor Cell Expansion Medium XF is a serum-free, xeno-free medium for expansion of primitive CD34+ HPCs. The medium, when combined with Cytokine Mix E, can support a 50-200-fold expansion of CD34+ progenitor cells.
- PromoExQ HPC Expansion Medium XF offers Excipient GMP-grade* quality with complete documentation to support scalable manufacturing and regulatory submissions.
These tools can help you expand primitive hematopoietic progenitor cells reliably, whether you’re optimizing basic protocols or advancing clinical applications.
*”GMP-grade” is a branding term used by PromoCell to denote reagents that are manufactured at the PromoCell manufacturing facility in Heidelberg, Germany, under strictly controlled processes to meet stringent product specifications and customer requirements. Reagents manufactured at PromoCell are produced according to EXCiPACT™ GMP standards, a quality management system that builds on our ISO 9001:2015 certification. Risk assessment procedures are carried out at the customer site.
Contact our experts
Contact our scientific support team to learn more about our solutions for CD34+ cell research and manufacturing and to discuss your research needs. Whether you’re investigating blood cell development, optimizing expansion protocols, or developing clinical cell therapies, we’re here to help you advance your research with the high-quality cells and media you need to succeed.

Figure 3: Disease context shapes macrophage function. Macrophage plasticity plays opposing roles in different diseases. In cancer, tumor progression promotes M2-like phenotypes that support growth and metastasis. In chronic inflammatory diseases, persistent M1 activation drives tissue damage. Therapeutic approaches aim to restore beneficial activation patterns.
References
Expand
- Sidney LE, Branch MJ, Dunphy SE, Dua HS, Hopkinson A. Concise review: Evidence for CD34 as a common marker for diverse progenitors. Stem Cells.. 2014;32(6):1380-1389. doi:10.1002/stem.1661
- Hughes MR, Canals Hernaez D, Cait J, et al. A sticky wicket: Defining molecular functions for CD34 in hematopoietic cells. Experimental Hematology.. 2020;86:1-14. doi:10.1016/j.exphem.2020.05.004
- Sadiq IZ, Abubakar FS, Katsayal BS, et al. Stem cells in regenerative medicine: Unlocking therapeutic potential through stem cell therapy, 3D bioprinting, gene editing, and drug discovery. Biomedical Engineering Advances.. 2025;9:100172. doi:10.1016/j.bea.2025.100172
- Pei X. Who is hematopoietic stem cell: CD34+ or CD34-? Int J Hematol. . 1999;70(4):213-215.
- AbuSamra DB, Aleisa FA, Al-Amoodi AS, et al. Not just a marker: CD34 on human hematopoietic stem/progenitor cells dominates vascular selectin binding along with CD44. Blood Adv. . 2017;1(27):2799-2816. doi:10.1182/bloodadvances.2017004317
- Schulz C, von Andrian UH, Massberg S. Hematopoietic stem and progenitor cells: their mobilization and homing to bone marrow and peripheral tissue. Immunol Res.. 2009;44(1-3):160-168. doi:10.1007/s12026-009-8109-6
- In brief: What does blood do? In: InformedHealth.Org [Internet]. Institute for Quality and Efficiency in Health Care (IQWiG). 2023. Accessed January 6, 2026. https://www.ncbi.nlm.nih.gov/books/NBK279392/
- Chen Z, Ju Z. Inflamm-aging of hematopoietic stem cells. Blood Sci.. 2019;1(2):141-143. doi:10.1097/BS9.0000000000000029
- Anjos-Afonso F, Bonnet D. Human CD34+ hematopoietic stem cell hierarchy: how far are we with its delineation at the most primitive level? Blood.. 2023;142(6):509-518. doi:10.1182/blood.2022018071
- Hassanpour M, Salybekov AA, Kobayashi S, Asahara T. CD34 positive cells as endothelial progenitor cells in biology and medicine. Front Cell Dev Biol.. 2023;11. doi:10.3389/fcell.2023.1128134
- Yu JT, Cheng SB, Yang Y, Chang KH, Hwang WL, Teng CLJ. Circulating hematopoietic progenitors and CD34+ cells predicted successful hematopoietic stem cell harvest in myeloma and lymphoma patients: experiences from a single institution. J Blood Med.. 2016;7:5-11. doi:10.2147/JBM.S95679
- Seita J, Weissman IL. Hematopoietic stem cell: self‐renewal versus differentiation. WIREs Mechanisms of Disease.. 2010;2(6):640-653. doi:10.1002/wsbm.86
- Yang J, Ii M, Kamei N, et al. CD34+ cells represent highly functional endothelial progenitor cells in murine bone marrow. Capogrossi MC, ed. PLoS ONE.. 2011;6(5):e20219. doi:10.1371/journal.pone.0020219
- Lee JY, Hong SH. Hematopoietic stem cells and their roles in tissue regeneration. Int J Stem Cells.. 2019;13(1):1-12. doi:10.15283/ijsc19127
- Seita J, Weissman IL. Hematopoietic stem cell: Self-renewal versus differentiation. Wiley Interdiscip Rev Syst Biol Med.. 2010;2(6):640-653. doi:10.1002/wsbm.86
- Kondo M. Lymphoid and myeloid lineage commitment in multipotent hematopoietic progenitors. Immunol Rev.. 2010;238(1):37-46. doi:10.1111/j.1600-065X.2010.00963.x
- Bozhilov YK, Hsu I, Brown EJ, Wilkinson AC. In vitro human haematopoietic stem cell expansion and differentiation. Cells.. 2023;12(6):896. doi:10.3390/cells12060896
- Tran BT, Jeyanathan V, Cao R, Kaufmann E, King KY. Hematopoietic stem and progenitor cells as a reservoir for trained immunity. eLife. . 2025;14:e106610. doi:10.7554/eLife.106610
- Schippel N, Sharma S. Dynamics of human hematopoietic stem and progenitor cell differentiation to the erythroid lineage. Experimental Hematology.. 2023;123:1-17. doi:10.1016/j.exphem.2023.05.001
- Drissen R, Buza-Vidas N, Woll P, et al. Distinct myeloid progenitor differentiation pathways identified through single cell RNA sequencing. Nat Immunol. . 2016;17(6):666-676. doi:10.1038/ni.3412
- Adelman ER, Huang HT, Roisman A, et al. Aging human Hematopoietic stem cells manifest profound epigenetic reprogramming of enhancers that may predispose to leukemia. Cancer Discov.. 2019;9(8):1080-1101. doi:10.1158/2159-8290.CD-18-1474
- Li J, Wang Y, Zheng X, et al. Efficient generation of CAR NK cells from human umbilical cord blood CD34+ stem and progenitors for democratizing affordable immunotherapy. bioRxiv.. Preprint posted online July 30, 2024:2024.07.30.605741. doi:10.1101/2024.07.30.605741
- Koenig L, Juglair L, Tao TP, et al. A microfluidic bone marrow chip for the safety profiling of biologics in pre-clinical drug development. Commun Biol.. 2025;8:754. doi:10.1038/s42003-025-08137-1
- Germino-Watnick P, Hinds M, Le A, Chu R, Liu X, Uchida N. Hematopoietic stem cell gene-addition/editing therapy in sickle cell disease. Cells.. 2022;11(11):1843. doi:10.3390/cells11111843
- Alavi A, Krishnamurti L, Abedi M, et al. Preliminary safety and efficacy results from Precizn-1: an ongoing phase 1/2 study on zinc finger nuclease-modified autologous CD34+ HSPCs for sickle cell disease (SCD). Blood.. 2021;138(Supplement 1):2930-2930. doi:10.1182/blood-2021-151650
- Holyoake TL, Alcorn MJ. CD34+ positive haemopoietic cells: biology and clinical applications. Blood Rev.. 1994;8(2):113-124. doi:10.1016/s0268-960x(05)80016-5
- Bhave R, Kath CJ, Rüchel N, et al. Serum-free differentiation platform for the generation of B lymphocytes and natural killer cells from human CD34+ cord blood progenitors. Sci Rep.. 2025;15(1):44132. doi:10.1038/s41598-025-30732-9
Related resources
